From a consideration of the phase diagram below,a change from point M to point N corresponds to 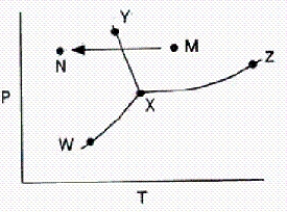
A) sublimation.
B) liquefaction.
C) evaporation.
D) condensation.
E) freezing.
Correct Answer:
Verified
Q17: Which of the following phase changes are
Q18: The enthalpy of fusion of aluminum is
Q19: Which of the following statements concerning liquids
Q20: How much heat is released at constant
Q21: Which explanation best describes surface tension?
A)Molecules at
Q23: The critical point of CCl4 is 283°C
Q24: The triple point of iodine is at
Q25: Choose the correct statement about the diagram
Q26: In which of the following substances are
Q27: If the liquid of a pure substance
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents