For the hypothetical reaction aA → products,the concentration of A was monitored with time.Given the following graph of the experimental data,what is the rate constant for the loss of reactant A? 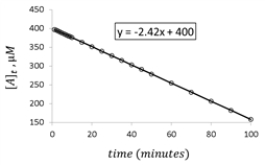
A) 2.42 μΜ −1⋅min−1
B) 400.00 μΜ −1⋅min−1
C) -2.42 μΜ −1⋅min−1
D) -400 μΜ −1⋅min−1
E) 24.2 μΜ −1⋅min−1
Correct Answer:
Verified
Q68: When the concentrations of the reactants are
Q69: The rates of most chemical reactions are
Q70: For a certain reaction of the general
Q71: The OH· radical disproportionates according to the
Q72: For a certain reaction of the general
Q74: The rate constant for a first-order reaction
Q75: For the hypothetical reaction A → products,the
Q76: In a first-order reaction,the half-life is 139
Q77: What would happen if the kinetic energy
Q78: For the formation of 1 mol of
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents