Deck 6: Reactions of Alkenes
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question

Unlock Deck
Sign up to unlock the cards in this deck!
Unlock Deck
Unlock Deck
1/79
Play
Full screen (f)
Deck 6: Reactions of Alkenes
1
What is the major organic product obtained from the following reaction? 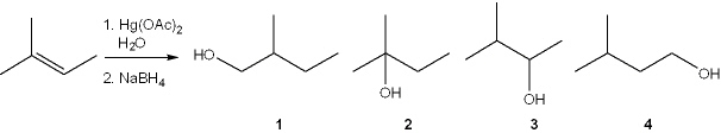
A) 1
B) 2
C) 3
D) 4

A) 1
B) 2
C) 3
D) 4
2
2
What type of carbocation is shown? 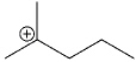
A) primary
B) secondary
C) tertiary
D) quaternary

A) primary
B) secondary
C) tertiary
D) quaternary
tertiary
3
What is the major organic product obtained from the following reaction? 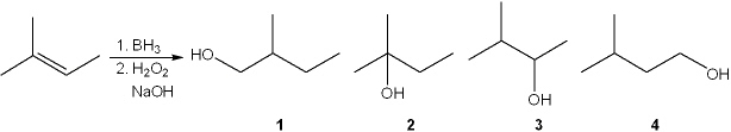
A) 1
B) 2
C) 3
D) 4

A) 1
B) 2
C) 3
D) 4
3
4
Which of the following alkenes is most likely to undergo rearrangement upon acid-catalyzed hydration (treatment with aqueous H2SO4)? 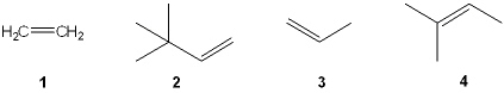
A) 1
B) 2
C) 3
D) 4

A) 1
B) 2
C) 3
D) 4

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
5
What is the major organic product obtained from the following reaction? 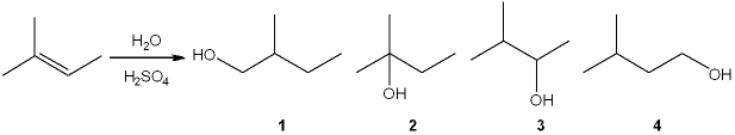
A) 1
B) 2
C) 3
D) 4

A) 1
B) 2
C) 3
D) 4

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
6
What is the major organic product obtained from the following reaction? 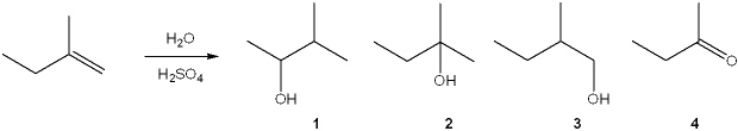
A) 1
B) 2
C) 3
D) 4

A) 1
B) 2
C) 3
D) 4

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
7
What is the correct order of stability of the following carbocations (more stable > less stable)? 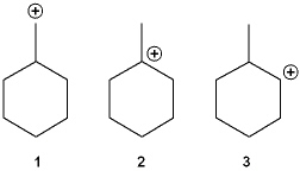
A) 1 > 2 > 3
B) 2 > 1 > 3
C) 2 > 3 > 1
D) 3 > 2 > 1

A) 1 > 2 > 3
B) 2 > 1 > 3
C) 2 > 3 > 1
D) 3 > 2 > 1

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
8
Which of the following concepts explains Markovnikov's rule as applied to the addition of HBr to propene?
A) the relative stability of carbocations
B) the nucleophilicity of bromide anion
C) the acidity of HBr
D) the Aufbau principle
A) the relative stability of carbocations
B) the nucleophilicity of bromide anion
C) the acidity of HBr
D) the Aufbau principle

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
9
What is the major organic product obtained from the following reaction? 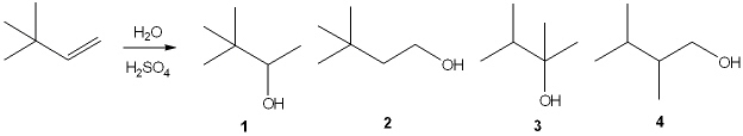
A) 1
B) 2
C) 3
D) 4

A) 1
B) 2
C) 3
D) 4

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
10
What type of carbocation is shown? 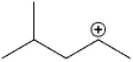
A) primary
B) secondary
C) tertiary
D) quaternary

A) primary
B) secondary
C) tertiary
D) quaternary

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
11
What is the major organic product obtained from the following reaction? 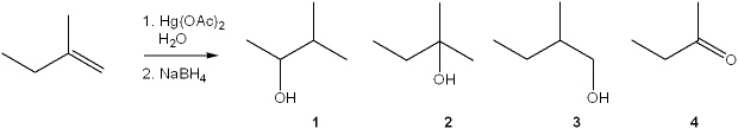
A) 1
B) 2
C) 3
D) 4

A) 1
B) 2
C) 3
D) 4

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
12
Which of the following concepts explains why tertiary carbocations are more stable than primary and secondary carbocations?
A) electronegativity
B) resonance
C) hyperconjugation
D) the octet rule
A) electronegativity
B) resonance
C) hyperconjugation
D) the octet rule

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
13
Which of the following alkenes is most likely to undergo rearrangement upon acid-catalyzed hydration (treatment with aqueous H2SO4)? 
A) 1
B) 2
C) 3
D) 4

A) 1
B) 2
C) 3
D) 4

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
14
What type of orbitals overlap to provide stability to the tert-butyl carbocation by hyperconjugation?
A) 3 C 2p atomic orbital + 3 C sp2 atomic orbital
B) 3 C 2p atomic orbital + methyl C-H s molecular orbital
C) 3 C sp2 atomic orbital + methyl C-H s molecular orbital
D) 3 C 2p atomic orbital + methyl C 2s atomic orbital
A) 3 C 2p atomic orbital + 3 C sp2 atomic orbital
B) 3 C 2p atomic orbital + methyl C-H s molecular orbital
C) 3 C sp2 atomic orbital + methyl C-H s molecular orbital
D) 3 C 2p atomic orbital + methyl C 2s atomic orbital

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
15
What is the major organic product obtained from the following reaction? 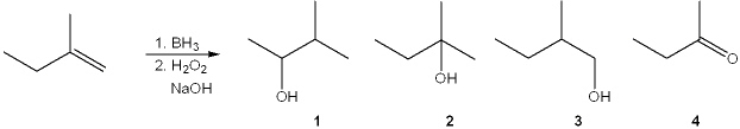
A) 1
B) 2
C) 3
D) 4

A) 1
B) 2
C) 3
D) 4

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
16
What is the major organic product obtained from the following reaction? 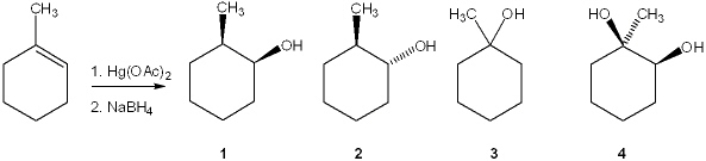
A) 1
B) 2
C) 3
D) 4

A) 1
B) 2
C) 3
D) 4

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
17
What is the major organic product obtained from the following reaction? 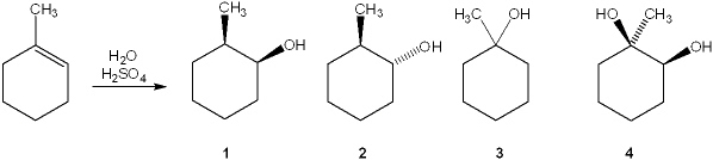
A) 1
B) 2
C) 3
D) 4

A) 1
B) 2
C) 3
D) 4

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
18
What is the major organic product obtained from the following reaction? 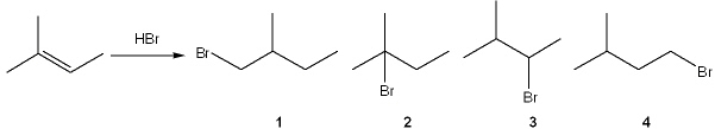
A) 1
B) 2
C) 3
D) 4

A) 1
B) 2
C) 3
D) 4

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
19
What is the major organic product obtained from the following reaction? 
A) 1
B) 2
C) 3
D) 4

A) 1
B) 2
C) 3
D) 4

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
20
What is the correct order of stability of the following carbocations (more stable > less stable)? 
A) 1 > 2 > 3
B) 1 > 3 > 2
C) 3 > 1 > 2
D) 3 > 2 > 1

A) 1 > 2 > 3
B) 1 > 3 > 2
C) 3 > 1 > 2
D) 3 > 2 > 1

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
21
What is the major organic product obtained from the following reaction? 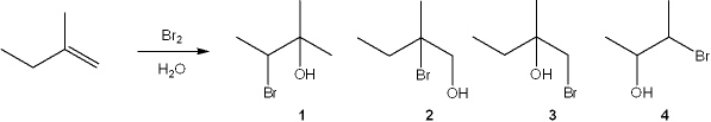
A) 1
B) 2
C) 3
D) 4

A) 1
B) 2
C) 3
D) 4

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
22
What is the best choice of reagent(s) to perform the following transformation? 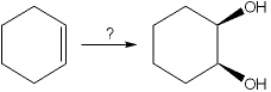
A) H2O, H2SO4
B) HgSO4; followed by NaBH4
C) BH3; followed by H2O2
D) OsO4; followed by NaHSO3

A) H2O, H2SO4
B) HgSO4; followed by NaBH4
C) BH3; followed by H2O2
D) OsO4; followed by NaHSO3

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
23
What is the major organic product obtained from the following reaction? 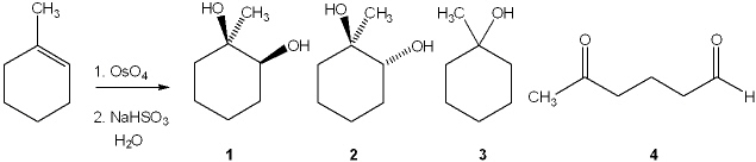
A) 1
B) 2
C) 3
D) 4

A) 1
B) 2
C) 3
D) 4

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
24
What is the major organic product obtained from the following reaction? 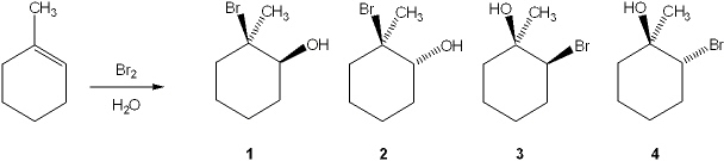
A) 1
B) 2
C) 3
D) 4

A) 1
B) 2
C) 3
D) 4

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
25
What is the major organic product obtained from the following reaction? 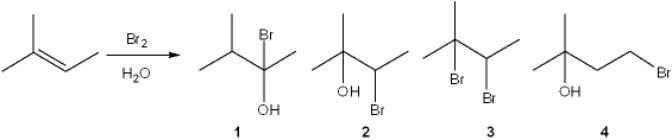
A) 1
B) 2
C) 3
D) 4

A) 1
B) 2
C) 3
D) 4

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
26
What is the major organic product obtained from the following reaction? 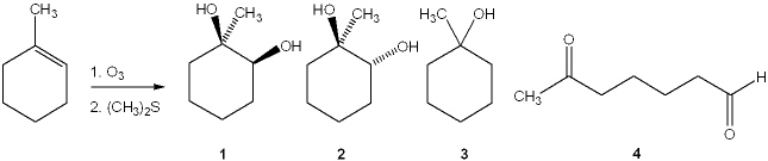
A) 1
B) 2
C) 3
D) 4

A) 1
B) 2
C) 3
D) 4

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
27
What is the best choice of reagent to perform the following transformation? 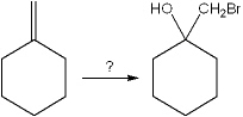
A) Br2
B) HBr
C) Br2, H2O
D) N-bromosuccinimide

A) Br2
B) HBr
C) Br2, H2O
D) N-bromosuccinimide

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
28
What is the best choice of reagent(s) to perform the following transformation? 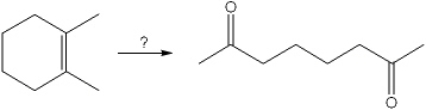
A) O3; followed by (CH3)2S
B) Hg(OAc)2 and H2O; followed by NaBH4
C) BH3; followed by H2O2, NaOH
D) OsO4; followed by NaHSO3

A) O3; followed by (CH3)2S
B) Hg(OAc)2 and H2O; followed by NaBH4
C) BH3; followed by H2O2, NaOH
D) OsO4; followed by NaHSO3

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
29
What is the major organic product obtained from the following reaction? 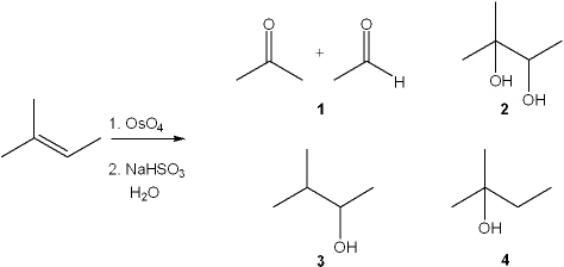
A) 1
B) 2
C) 3
D) 4

A) 1
B) 2
C) 3
D) 4

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
30
What is (are) the major organic product(s) obtained from the following reaction? 
A) only 1
B) only 2
C) only 3
D) only 1 and 2

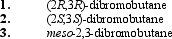
A) only 1
B) only 2
C) only 3
D) only 1 and 2

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
31
What type of reactive intermediate is formed in the reaction of an alkene with HBr to give a bromoalkane?
A) carbocation
B) carbanion
C) radical
D) cyclic bromonium ion
A) carbocation
B) carbanion
C) radical
D) cyclic bromonium ion

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
32
What is (are) the major organic product(s) obtained from the following reaction? 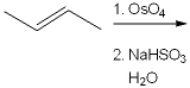

A) only 1
B) only 2
C) only 3
D) only 1 and 2


A) only 1
B) only 2
C) only 3
D) only 1 and 2

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
33
What is the major organic product obtained from the following reaction? 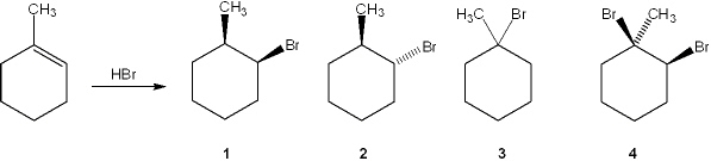
A) 1
B) 2
C) 3
D) 4

A) 1
B) 2
C) 3
D) 4

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
34
What is (are) the major organic product(s) obtained from the following reaction? 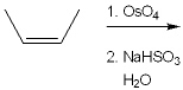

A) only 1
B) only 2
C) only 3
D) only 1 and 2
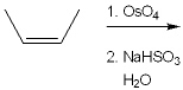

A) only 1
B) only 2
C) only 3
D) only 1 and 2

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
35
What is (are) the major organic product(s) obtained from the following reaction? 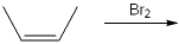
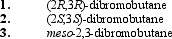
A) only 1
B) only 2
C) only 3
D) only 1 and 2
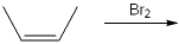
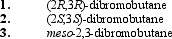
A) only 1
B) only 2
C) only 3
D) only 1 and 2

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
36
What is (are) the major organic product(s) obtained from the following reaction? 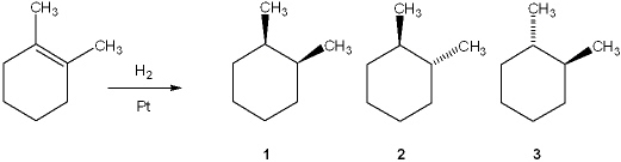
A) only 1
B) only 2
C) only 3
D) only 2 and 3

A) only 1
B) only 2
C) only 3
D) only 2 and 3

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
37
What is the best choice of reagent(s) to perform the following transformation? 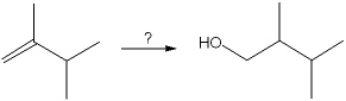
A) H2O, H2SO4
B) Hg(OAc)2 and H2O ; followed by NaBH4
C) B2H6; followed by H2O2, NaOH
D) OsO4; followed by NaHSO3

A) H2O, H2SO4
B) Hg(OAc)2 and H2O ; followed by NaBH4
C) B2H6; followed by H2O2, NaOH
D) OsO4; followed by NaHSO3

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
38
What is the major organic product obtained from the following reaction? 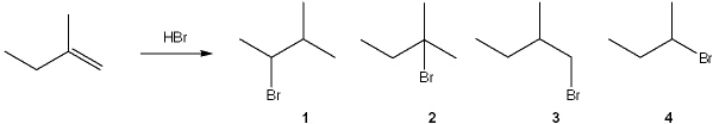
A) 1
B) 2
C) 3
D) 4

A) 1
B) 2
C) 3
D) 4

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
39
What is the best choice of reagent(s) to perform the following transformation? 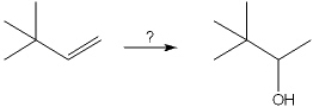
A) H2O, H2SO4
B) Hg(OAc)2 and H2O; followed by NaBH4
C) BH3; followed by H2O2, NaOH
D) OsO4; followed by NaHSO3

A) H2O, H2SO4
B) Hg(OAc)2 and H2O; followed by NaBH4
C) BH3; followed by H2O2, NaOH
D) OsO4; followed by NaHSO3

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
40
What is the major product obtained upon addition of Br2 to (R)-4-tert-butylcyclohexene?
A) (1R,2R,4R)-1,2-dibromo-4-tert-butylcyclohexane
B) (1S,2R,4R)-1,2-dibromo-4-tert-butylcyclohexane
C) (1S,2S,4R)-1,2-dibromo-4-tert-butylcyclohexane
D) (1S,2S,4S)-1,2-dibromo-4-tert-butylcyclohexane
A) (1R,2R,4R)-1,2-dibromo-4-tert-butylcyclohexane
B) (1S,2R,4R)-1,2-dibromo-4-tert-butylcyclohexane
C) (1S,2S,4R)-1,2-dibromo-4-tert-butylcyclohexane
D) (1S,2S,4S)-1,2-dibromo-4-tert-butylcyclohexane

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
41
Which of the following reactions of alkenes takes place with syn stereospecificity?
A) addition of bromine (treatment with Br2)
B) hydrogenation (treatment with H2/Pt)
C) addition of HBr (treatment with HBr)
D) acid-catalyzed hydration (treatment with aqueous H2SO4)
A) addition of bromine (treatment with Br2)
B) hydrogenation (treatment with H2/Pt)
C) addition of HBr (treatment with HBr)
D) acid-catalyzed hydration (treatment with aqueous H2SO4)

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
42
Which of the following alkenes undergoes the least exothermic hydrogenation upon treatment with H2/Pd? 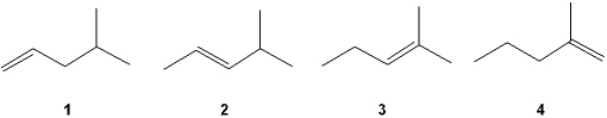
A) 1
B) 2
C) 3
D) 4

A) 1
B) 2
C) 3
D) 4

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
43
What is (are) the major organic product(s) obtained from the following reaction? 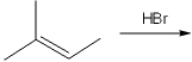

A) only 1
B) only 2
C) only 3
D) only 1 and 2


A) only 1
B) only 2
C) only 3
D) only 1 and 2

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
44
What is the major organic product obtained from the following reaction?



Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
45
What is the correct order of exothermicity for hydrogenation of the following butenes upon treatment with H2/Pd (more exothermic > less exothermic)? 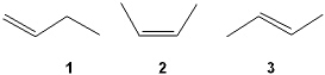
A) 1 > 2 > 3
B) 1 > 3 > 2
C) 3 > 2 > 1
D) 2 > 3 > 1

A) 1 > 2 > 3
B) 1 > 3 > 2
C) 3 > 2 > 1
D) 2 > 3 > 1

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
46
What type of reactive intermediate is formed in the reaction of an alkene with aqueous acid to give an alcohol?
A) carbocation
B) carbanion
C) radical
D) carbene
A) carbocation
B) carbanion
C) radical
D) carbene

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
47
What is the major organic product obtained from the following reaction?



Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
48
What is the major organic product obtained from the following reaction?



Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
49
What is the major organic product obtained from the following reaction?
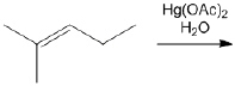


Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
50
What is the major organic product obtained from the following reaction?
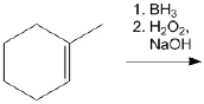


Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
51
Which of the following reactions of alkenes is not stereospecific?
A) bromination (treatment with Br2 in CHCl3)
B) hydrogenation (treatment with H2/Pt)
C) acid-catalyzed hydration (treatment with aqueous H2SO4)
D) bromohydrin formation (treatment with Br2/H2O)
A) bromination (treatment with Br2 in CHCl3)
B) hydrogenation (treatment with H2/Pt)
C) acid-catalyzed hydration (treatment with aqueous H2SO4)
D) bromohydrin formation (treatment with Br2/H2O)

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
52
What is the major organic product obtained from the following reaction?
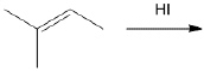


Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
53
What is (are) the major organic product(s) obtained from the following reaction? 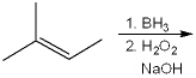

A) only 1
B) only 2
C) only 3
D) only 1 and 2


A) only 1
B) only 2
C) only 3
D) only 1 and 2

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
54
What is the major organic product obtained from the following reaction?
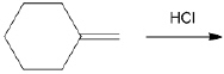


Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
55
Which of the following reactions of alkenes is stereospecific?
A) addition of HCl (treatment with HCl)
B) hydrogenation (treatment with H2/Pt)
C) addition of HBr (treatment with HBr)
D) acid-catalyzed hydration (treatment with aqueous H2SO4)
A) addition of HCl (treatment with HCl)
B) hydrogenation (treatment with H2/Pt)
C) addition of HBr (treatment with HBr)
D) acid-catalyzed hydration (treatment with aqueous H2SO4)

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
56
Which of the following alkenes undergoes the most exothermic hydrogenation upon treatment with H2/Pd? 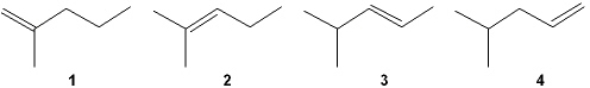
A) 1
B) 2
C) 3
D) 4

A) 1
B) 2
C) 3
D) 4

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
57
What type of reaction mechanism accounts for the reaction of an alkene with HBr to give an alkyl bromide?
A) nucleophilic addition
B) electrophilic addition
C) radical addition
D) elimination
A) nucleophilic addition
B) electrophilic addition
C) radical addition
D) elimination

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
58
What type of reactive intermediate is formed in the reaction of an alkene with Br2 and water to give a bromohydrin?
A) carbocation
B) carbanion
C) radical
D) cyclic bromonium ion
A) carbocation
B) carbanion
C) radical
D) cyclic bromonium ion

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
59
Which of the following reactions of alkenes takes place with anti stereospecificity?
A) bishydroxylation (treatment with OsO4 followed by NaHSO3)
B) hydrogenation (treatment with H2/Pt)
C) addition of HBr (treatment with HBr)
D) bromohydrin formation (treatment with Br2, H2O)
A) bishydroxylation (treatment with OsO4 followed by NaHSO3)
B) hydrogenation (treatment with H2/Pt)
C) addition of HBr (treatment with HBr)
D) bromohydrin formation (treatment with Br2, H2O)

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
60
What type of reaction mechanism accounts for the reaction of an alkene with aqueous acid to give an alcohol?
A) nucleophilic addition
B) electrophilic addition
C) radical addition
D) elimination
A) nucleophilic addition
B) electrophilic addition
C) radical addition
D) elimination

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
61
What is the best choice of reagent(s) to perform the following transformation?
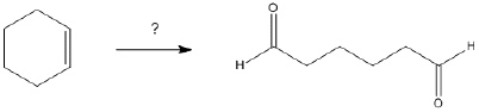


Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
62
Provide a neatly drawn mechanism for the following reaction, including curved arrows to show the movement of paris of electrons and the structure of reactive intermediates. 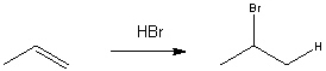 The reaction proceeds in two steps:
The reaction proceeds in two steps:
1. Protonation of propene to form the 2-propyl cation
2. Nucleophilic addition of bromide anion to the 2-propyl cation
 The reaction proceeds in two steps:
The reaction proceeds in two steps:1. Protonation of propene to form the 2-propyl cation
2. Nucleophilic addition of bromide anion to the 2-propyl cation

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
63
What is the best choice of reagent(s) to perform the following transformation?
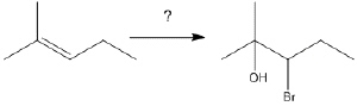


Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
64
Provide a neatly drawn mechanism for the following reaction, including curved arrows to show the movement of paris of electrons and the structure of reactive intermediates. 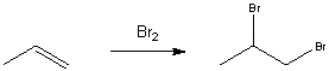 The reaction proceeds in two steps:
The reaction proceeds in two steps:
1. Addition of bromine to propene to afford a cyclic bromonium ion
2. Nucleophilic ring-opening addition of bromide anion
 The reaction proceeds in two steps:
The reaction proceeds in two steps:1. Addition of bromine to propene to afford a cyclic bromonium ion
2. Nucleophilic ring-opening addition of bromide anion

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
65
What is the best choice of reagent(s) to perform the following transformation?
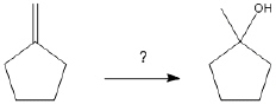


Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
66
Provide the structure of the key intermediate in the following reaction?
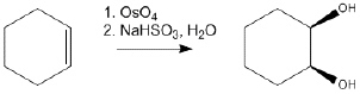


Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
67
What is the major organic product obtained from the following reaction?
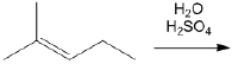


Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
68
Provide the structure of the key intermediate in the following reaction?
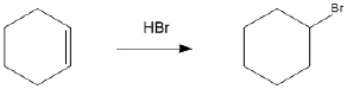


Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
69
What is the major organic product obtained from the following reaction?
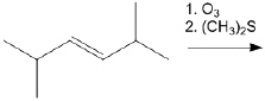 2 equivalents of
2 equivalents of
 2 equivalents of
2 equivalents of 
Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
70
Provide a neatly drawn mechanism for the following reaction, including curved arrows to show the movement of paris of electrons and the structure of reactive intermediates.

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
71
What is the major organic product obtained from the following reaction?
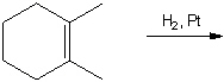


Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
72
Provide the structure of the key intermediate in the following reaction?
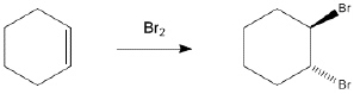


Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
73
Provide a neatly drawn mechanism for the following reaction, including curved arrows to show the movement of paris of electrons and the structure of reactive intermediates. 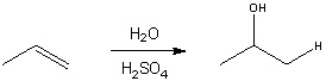 The reaction proceeds in three steps:
The reaction proceeds in three steps:
1. Protonation of propene to form the 2-propyl cation
2. Nucleophilic addition of bromine to the 2-propyl cation
3. Deprotonation of the oxygen
 The reaction proceeds in three steps:
The reaction proceeds in three steps:1. Protonation of propene to form the 2-propyl cation
2. Nucleophilic addition of bromine to the 2-propyl cation
3. Deprotonation of the oxygen

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
74
What is the best choice of reagent(s) to perform the following transformation?
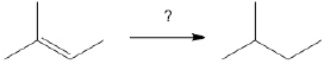


Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
75
What is the best choice of reagent(s) to perform the following transformation?

Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
76
What is the best choice of reagent(s) to perform the following transformation?
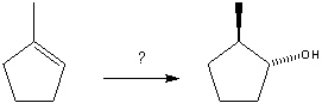


Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
77
What is the best choice of reagent(s) to perform the following transformation?
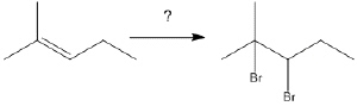


Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
78
What is the major organic product obtained from the following reaction?
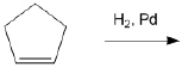


Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck
79
What is the best choice of reagent(s) to perform the following transformation?
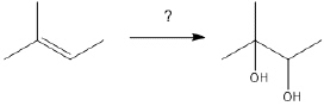


Unlock Deck
Unlock for access to all 79 flashcards in this deck.
Unlock Deck
k this deck



