Deck 17: Entropy Free Energy and Equilibrium
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question

Unlock Deck
Sign up to unlock the cards in this deck!
Unlock Deck
Unlock Deck
1/128
Play
Full screen (f)
Deck 17: Entropy Free Energy and Equilibrium
1
Which of these species has the highest entropy (S°)at 25°C?
A)CH3OH(l)
B)CO(g)
C)MgCO3(s)
D)H2O(l)
E)Ni(s)
A)CH3OH(l)
B)CO(g)
C)MgCO3(s)
D)H2O(l)
E)Ni(s)
CO(g)
2
Which of the following processes would be accompanied by an increase in entropy?
A)H2O(g) H2O(s)
B)H2O(l) H2O(s)
C)H2O(g) H2O(l)
D)H2O(s) H2O(g)
E)H2O(l, 50°C) H2O(l, 25°C)
A)H2O(g) H2O(s)
B)H2O(l) H2O(s)
C)H2O(g) H2O(l)
D)H2O(s) H2O(g)
E)H2O(l, 50°C) H2O(l, 25°C)
H2O(s) H2O(g)
3
Which response includes all of the following processes that are accompanied by an increase in entropy?
1)I2(s) I2(g)
2)2I(g) I2(g)
3)2NH3(g) N2(g)+ 3H2(g)
4)Mg2+(aq)+ 2OH-(aq) Mg(OH)2(s)
A)1, 2
B)1, 3
C)3, 4
D)3
E)2, 4
1)I2(s) I2(g)
2)2I(g) I2(g)
3)2NH3(g) N2(g)+ 3H2(g)
4)Mg2+(aq)+ 2OH-(aq) Mg(OH)2(s)
A)1, 2
B)1, 3
C)3, 4
D)3
E)2, 4
1, 3
4
Aluminum forms a layer of aluminum oxide when exposed to air which protects the bulk metal from further corrosion. 4Al(s)+ 3O2(g) 2Al2O3(s)
Using the thermodynamic data provided below, calculate S° for this reaction.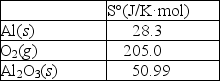
A)182.3 J/K·mol
B)131.5 J/K·mol
C)-182.3 J/K·mol
D)-626.2 J/K·mol
E)-802.9 J/K·mol
Using the thermodynamic data provided below, calculate S° for this reaction.

A)182.3 J/K·mol
B)131.5 J/K·mol
C)-182.3 J/K·mol
D)-626.2 J/K·mol
E)-802.9 J/K·mol

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
5
Which of the following is expected to have zero entropy? I.N2(g)at 273 K
II.SiO2(s, amorphous)at 0 K
III.NaCl(s)perfectly ordered crystal at 25 K
IV.Na(s)perfectly ordered crystal at 0 K
A)I and IV
B)III and IV
C)I and II
D)I, II, and III
E)IV only
II.SiO2(s, amorphous)at 0 K
III.NaCl(s)perfectly ordered crystal at 25 K
IV.Na(s)perfectly ordered crystal at 0 K
A)I and IV
B)III and IV
C)I and II
D)I, II, and III
E)IV only

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
6
Which of these species would you expect to have the highest standard entropy (S°)?
A)CH4(g)
B)C2H2(g)
C)C2H4(g)
D)C2H6(g)
E)C3H8(g)
A)CH4(g)
B)C2H2(g)
C)C2H4(g)
D)C2H6(g)
E)C3H8(g)

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
7
Which of these species has the highest entropy (S°)at 25°C?
A)CO(g)
B)CH4(g)
C)NaCl(s)
D)H2O(l)
E)Fe(s)
A)CO(g)
B)CH4(g)
C)NaCl(s)
D)H2O(l)
E)Fe(s)

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
8
Without reference to a table, arrange these reactions according to increasing S. 1)CH4(g)+ H2O(g) CO(g)+ 3H2(g)
2)C(s)+ O2(g) CO2(g)
3)H2O2(l) H2O(l)+ 1/2O2(g)
A)1 < 3 < 2
B)2 < 3 < 1
C)2 < 1 < 3
D)3 < 2 < 1
E)3 < 1 < 2
2)C(s)+ O2(g) CO2(g)
3)H2O2(l) H2O(l)+ 1/2O2(g)
A)1 < 3 < 2
B)2 < 3 < 1
C)2 < 1 < 3
D)3 < 2 < 1
E)3 < 1 < 2

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
9
Arrange the following substances in the order of increasing entropy at 25°C. HF(g), NaF(s), SiF4(g), SiH4(g), Al(s)
Lowest highest
A)SiF4(g)< SiH4(g)< NaF(s)< HF(g)< Al(s)
B)HF(g)< Al(s)< NaF(s)< SiF4(g)< SiH4(g)
C)Al(s)< NaF(s)< HF(g)< SiH4(g)< SiF4(g)
D)Al(s)< HF(g)< NaF(s)< SiF4(g)< SiH4(g)
E)NaF(s)< Al(s)< HF(g)< SiF4(g)< SiH4(g)
Lowest highest
A)SiF4(g)< SiH4(g)< NaF(s)< HF(g)< Al(s)
B)HF(g)< Al(s)< NaF(s)< SiF4(g)< SiH4(g)
C)Al(s)< NaF(s)< HF(g)< SiH4(g)< SiF4(g)
D)Al(s)< HF(g)< NaF(s)< SiF4(g)< SiH4(g)
E)NaF(s)< Al(s)< HF(g)< SiF4(g)< SiH4(g)

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
10
Which one of the following reactions would you expect to have highest S°?
A)CH4(g)+ 2O2(g) CO2(g)+ 2H2O(g)
B)C2H2(g)+ 5/2O2(g) 2CO2(g)+ H2O(g)
C)C2H4(g)+ 3O2(g) 2CO2(g)+ 2H2O(g)
D)C2H6(g)+ 7/2O2(g) 2CO2(g)+ 3H2O(g)
A)CH4(g)+ 2O2(g) CO2(g)+ 2H2O(g)
B)C2H2(g)+ 5/2O2(g) 2CO2(g)+ H2O(g)
C)C2H4(g)+ 3O2(g) 2CO2(g)+ 2H2O(g)
D)C2H6(g)+ 7/2O2(g) 2CO2(g)+ 3H2O(g)

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
11
Arrange these reactions according to increasing S.
1)H2O(g) H2O(l)
2)2NO(g) N2(g)+ O2(g)
3)MgCO3(s) MgO(s)+ CO2(g)
A)1 < 2 < 3
B)2 < 3 < 1
C)3 < 2 < 1
D)2 < 1 < 3
E)1 < 3 < 2
1)H2O(g) H2O(l)
2)2NO(g) N2(g)+ O2(g)
3)MgCO3(s) MgO(s)+ CO2(g)
A)1 < 2 < 3
B)2 < 3 < 1
C)3 < 2 < 1
D)2 < 1 < 3
E)1 < 3 < 2

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
12
Which of these species would you expect to have the lowest standard entropy (S°)?
A)CH4(g)
B)HF(g)
C)NH3(g)
D)H2O(g)
A)CH4(g)
B)HF(g)
C)NH3(g)
D)H2O(g)

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
13
Which one of the following reactions would you expect to have the lowest S°?
A)CH4(g)+ 2O2(g) CO2(g)+ 2H2O(g)
B)C2H2(g)+ 5/2O2(g) 2CO2(g)+ H2O(g)
C)C2H4(g)+ O2(g) 2CO2(g)+ 2H2O(g)
D)C2H6(g)+ 7/2O2(g) 2CO2(g)+ 3H2O(g)
A)CH4(g)+ 2O2(g) CO2(g)+ 2H2O(g)
B)C2H2(g)+ 5/2O2(g) 2CO2(g)+ H2O(g)
C)C2H4(g)+ O2(g) 2CO2(g)+ 2H2O(g)
D)C2H6(g)+ 7/2O2(g) 2CO2(g)+ 3H2O(g)

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
14
Which response includes all of the following processes that are accompanied by an increase of entropy? 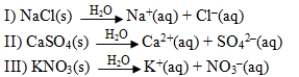
A)I and III
B)II and III
C)I, II and III
D)II only
E)III only
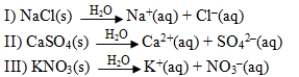
A)I and III
B)II and III
C)I, II and III
D)II only
E)III only

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
15
Determine S° for the reaction SO3(g)+ H2O(l) H2SO4(l). 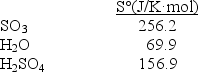
A)169.2 J/K·mol
B)1343.2 J/K·mol
C)-169.2 J/K·mol
D)-29.4 J/K·mol
E)29.4 J/K·mol
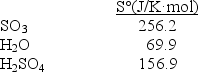
A)169.2 J/K·mol
B)1343.2 J/K·mol
C)-169.2 J/K·mol
D)-29.4 J/K·mol
E)29.4 J/K·mol

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
16
Arrange these compounds in order of increasing standard molar entropy at 25°C: C3H8(g), C2H4(g), ZnS(s), and H2O(l).
A)ZnS(s)< H2O(l)< C3H8(g)< C2H4(g)
B)C2H4(g)< H2O(l)< C3H8(g)< NaCl(s)
C)ZnS(s)< C3H8(g)< C2H4(g)< H2O(l)
D)C3H8(g)< C2H4(g)< H2O(l)< ZnS(s)
E)ZnS(s)< H2O(l)< C2H4(g)< C3H8(g)
A)ZnS(s)< H2O(l)< C3H8(g)< C2H4(g)
B)C2H4(g)< H2O(l)< C3H8(g)< NaCl(s)
C)ZnS(s)< C3H8(g)< C2H4(g)< H2O(l)
D)C3H8(g)< C2H4(g)< H2O(l)< ZnS(s)
E)ZnS(s)< H2O(l)< C2H4(g)< C3H8(g)

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
17
Sulfur can be separated from lead in the mineral galena, PbS(s), by "roasting" the ore in the presence of oxygen as shown in the following reaction:
2PbS(s)+ 3O2(g) 2PbO(s)+ 2SO2(g)
Calculate S° for this reaction using the thermodynamic data provided below.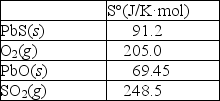
A)-410 J/K·mol
B)-161.5 J/K·mol
C)-47.7 J/K·mol
D)21.8 J/K·mol
E)43.5 J/K·mol
2PbS(s)+ 3O2(g) 2PbO(s)+ 2SO2(g)
Calculate S° for this reaction using the thermodynamic data provided below.

A)-410 J/K·mol
B)-161.5 J/K·mol
C)-47.7 J/K·mol
D)21.8 J/K·mol
E)43.5 J/K·mol

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
18
Which of these species would you expect to have the lowest standard entropy (S°)?
A)Br2(l)
B)Cl2(g)
C)F2(g)
D)H2(g)
E)I2(s)
A)Br2(l)
B)Cl2(g)
C)F2(g)
D)H2(g)
E)I2(s)

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
19
Which of the following processes would be accompanied by a decrease in entropy?
A)H2O(s) H2O(g)
B)H2O(l) H2O(g)
C)H2O(s) H2O(l)
D)H2O(g) H2O(s)
E)H2O(l, 50°C) H2O(l, 75°C)
A)H2O(s) H2O(g)
B)H2O(l) H2O(g)
C)H2O(s) H2O(l)
D)H2O(g) H2O(s)
E)H2O(l, 50°C) H2O(l, 75°C)

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
20
Which response includes all the following processes that are accompanied by an increase in entropy? 1)2SO2(g)+ O2(g) SO3(g)
2)H2O(l) H2O(s)
3)Br2(l) Br2(g)
4)H2O2(l) H2O(l)+ 1/2O2(g)
A)1, 2, 3, 4
B)1, 2
C)2, 3, 4
D)3, 4
E)1, 4
2)H2O(l) H2O(s)
3)Br2(l) Br2(g)
4)H2O2(l) H2O(l)+ 1/2O2(g)
A)1, 2, 3, 4
B)1, 2
C)2, 3, 4
D)3, 4
E)1, 4

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
21
Which of the following is consistent with an exothermic reaction that is nonspontaneous at high temperatures?
A)( H > 0, S < 0, G < 0)
B)( H > 0, S > 0, G < 0)
C)( H < 0, S > 0, G < 0)
D)( H < 0, S > 0, G > 0)
E)( H < 0, S < 0, G > 0)
A)( H > 0, S < 0, G < 0)
B)( H > 0, S > 0, G < 0)
C)( H < 0, S > 0, G < 0)
D)( H < 0, S > 0, G > 0)
E)( H < 0, S < 0, G > 0)

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
22
Ozone (O3)in the atmosphere can reaction with nitric oxide (NO): O3(g)+ NO(g) NO2(g)+ O2(g).
Calculate the G° for this reaction at 25°C.( H° = -199 kJ/mol, S° = -4.1 J/K·mol)
A)1020 kJ/mol
B)-1.22 × 103 kJ/mol
C)2.00 × 103 kJ/mol
D)-1.42 × 103 kJ/mol
E)-198 kJ/mol
Calculate the G° for this reaction at 25°C.( H° = -199 kJ/mol, S° = -4.1 J/K·mol)
A)1020 kJ/mol
B)-1.22 × 103 kJ/mol
C)2.00 × 103 kJ/mol
D)-1.42 × 103 kJ/mol
E)-198 kJ/mol

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
23
The normal melting point sulfur is 113 °C.If a sample of solid sulfur is at 95 °C, Predict the signs of H, S, and G for the melting process at this temperature.
A)( H > 0, S > 0, G < 0)
B)( H > 0, S > 0, G > 0)
C)( H > 0, S < 0, G < 0)
D)( H < 0, S > 0, G > 0)
E)( H < 0, S < 0, G > 0)
A)( H > 0, S > 0, G < 0)
B)( H > 0, S > 0, G > 0)
C)( H > 0, S < 0, G < 0)
D)( H < 0, S > 0, G > 0)
E)( H < 0, S < 0, G > 0)

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
24
Calculate G° for the reaction 3NO2(g)+ H2O(l) 2HNO3(l)+ NO(g). 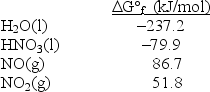
A)8.7 kJ/mol
B)192 kJ/mol
C)-414 kJ/mol
D)-192 kJ/mol
E)-155 kJ/mol
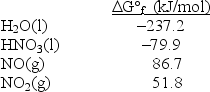
A)8.7 kJ/mol
B)192 kJ/mol
C)-414 kJ/mol
D)-192 kJ/mol
E)-155 kJ/mol

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
25
Sodium carbonate can be made by heating sodium bicarbonate: 2NaHCO3(s) Na2CO3(s)+ CO2(g)+ H2O(g)
Given that H° = 128.9 kJ/mol and G° = 33.1 kJ/mol at 25°C, above what minimum temperature will the reaction become spontaneous under standard state conditions?
A)0.4 K
B)3.9 K
C)321 K
D)401 K
E)525 K
Given that H° = 128.9 kJ/mol and G° = 33.1 kJ/mol at 25°C, above what minimum temperature will the reaction become spontaneous under standard state conditions?
A)0.4 K
B)3.9 K
C)321 K
D)401 K
E)525 K

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
26
Aluminum forms a layer of aluminum oxide when exposed to air which protects the bulk metal from further corrosion. 4Al(s)+ 3O2(g) 2Al2O3(s)
Calculate G° for this reaction, given that G°f of aluminum oxide is -1576.4 kJ/mol.
A)-3152.8 kJ/mol
B)-1576.4 kJ/mol
C)-788.2 kJ/mol
D)1576.4 kJ/mol
E)3152.8 kJ/mol
Calculate G° for this reaction, given that G°f of aluminum oxide is -1576.4 kJ/mol.
A)-3152.8 kJ/mol
B)-1576.4 kJ/mol
C)-788.2 kJ/mol
D)1576.4 kJ/mol
E)3152.8 kJ/mol

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
27
Which of the following is consistent with an exothermic reaction that is spontaneous at all temperatures?
A)( H > 0, S < 0, G < 0)
B)( H > 0, S > 0, G < 0)
C)( H < 0, S < 0, G < 0)
D)( H < 0, S > 0, G < 0)
E)( H > 0, S < 0, G > 0)
A)( H > 0, S < 0, G < 0)
B)( H > 0, S > 0, G < 0)
C)( H < 0, S < 0, G < 0)
D)( H < 0, S > 0, G < 0)
E)( H > 0, S < 0, G > 0)

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
28
Calculate S° for the reaction SO2(s)+ NO2(g) SO3(g)+ NO(g). 
A)53.6 J/K·mol
B)-53.6 J/K·mol
C)-22.2 J/K·mol
D)474.8 J/K·mol
E)-474.8 J/K·mol

A)53.6 J/K·mol
B)-53.6 J/K·mol
C)-22.2 J/K·mol
D)474.8 J/K·mol
E)-474.8 J/K·mol

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
29
The element oxygen was prepared by Joseph Priestley in 1774 by heating mercury(II)oxide: HgO(s) Hg(l)+ 1/2O2(g), H° = 90.84 kJ/mol.
Estimate the temperature at which this reaction will become spontaneous under standard state conditions.
S°(Hg)= 76.02 J/K·mol
S°(O2)= 205.0 J/K·mol
S°(HgO)= 70.29 J/K·mol
A)108 K
B)430 K
C)620 K
D)775 K
E)840 K
Estimate the temperature at which this reaction will become spontaneous under standard state conditions.
S°(Hg)= 76.02 J/K·mol
S°(O2)= 205.0 J/K·mol
S°(HgO)= 70.29 J/K·mol
A)108 K
B)430 K
C)620 K
D)775 K
E)840 K

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
30
The normal freezing point of ammonia is -78°C.Predict the signs of H, S, and G for ammonia when it freezes at -80°C and 1 atm: NH3(l) NH3(s) 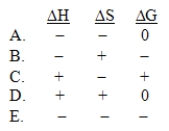
A)A
B)B
C)C
D)D
E)E

A)A
B)B
C)C
D)D
E)E

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
31
Which of the following is consistent with a spontaneous endothermic reaction?
A)( H > 0, S < 0, G < 0)
B)( H > 0, S > 0, G < 0)
C)( H < 0, S < 0, G < 0)
D)( H < 0, S > 0, G > 0)
E)( H > 0, S < 0, G > 0)
A)( H > 0, S < 0, G < 0)
B)( H > 0, S > 0, G < 0)
C)( H < 0, S < 0, G < 0)
D)( H < 0, S > 0, G > 0)
E)( H > 0, S < 0, G > 0)

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
32
The normal boiling point of acetic acid is 118.1°C.If a sample of the acetic acid is at 125.2°C, Predict the signs of H, S, and G for the boiling process at this temperature.
A)( H > 0, S > 0, G < 0)
B)( H > 0, S > 0, G > 0)
C)( H > 0, S < 0, G < 0)
D)( H < 0, S > 0, G > 0)
E)( H < 0, S < 0, G > 0)
A)( H > 0, S > 0, G < 0)
B)( H > 0, S > 0, G > 0)
C)( H > 0, S < 0, G < 0)
D)( H < 0, S > 0, G > 0)
E)( H < 0, S < 0, G > 0)

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
33
Hydrogen peroxide (H2O2)decomposes according to the equation H2O2(l) H2O(l)+ 1/2O2(g).
Calculate Kp for this reaction at 25°C.( H° = -98.2 kJ/mol, S° = 70.1 J/K·mol)
A)1.3 × 10-21
B)20.9
C)3.46 × 1017
D)7.5 × 1020
E)8.6 × 104
Calculate Kp for this reaction at 25°C.( H° = -98.2 kJ/mol, S° = 70.1 J/K·mol)
A)1.3 × 10-21
B)20.9
C)3.46 × 1017
D)7.5 × 1020
E)8.6 × 104

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
34
With respect to the system only, a reaction with H < 0 and S > 0 is predicted to be:
A)Spontaneous at all temperatures
B)Spontaneous at high temperatures only
C)Spontaneous at low temperatures only
D)Nonspontaneous at all temperatures
A)Spontaneous at all temperatures
B)Spontaneous at high temperatures only
C)Spontaneous at low temperatures only
D)Nonspontaneous at all temperatures

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
35
Calculate S° at 25°C for the reduction of PbO(s), 2PbO(s)+ C(s) 2Pb(s)+ CO2(g)given these absolute entropies: 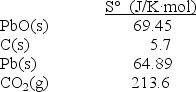
A)+198.8 J/K·mol
B)+488.0 J/K·mol
C)+353.6 J/K·mol
D)-203.3 J/K·mol
E)+203.3 J/K·mol
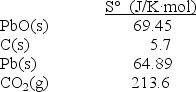
A)+198.8 J/K·mol
B)+488.0 J/K·mol
C)+353.6 J/K·mol
D)-203.3 J/K·mol
E)+203.3 J/K·mol

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
36
With respect to the system only, a reaction with H > 0 and S < 0 is predicted to be:
A)Spontaneous at all temperatures
B)Spontaneous at high temperatures only
C)Spontaneous at low temperatures only
D)Nonspontaneous at all temperatures
A)Spontaneous at all temperatures
B)Spontaneous at high temperatures only
C)Spontaneous at low temperatures only
D)Nonspontaneous at all temperatures

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
37
Which of the following is consistent with a nonspontaneous process in the forward direction?
A)( Suniv > 0, G < 0, T Suniv > 0)
B)( Suniv < 0, G > 0, T Suniv < 0)
C)( Suniv > 0, G < 0, T Suniv < 0)
D)( Suniv < 0, G < 0, T Suniv < 0)
E)( Suniv > 0, G > 0, T Suniv > 0)
A)( Suniv > 0, G < 0, T Suniv > 0)
B)( Suniv < 0, G > 0, T Suniv < 0)
C)( Suniv > 0, G < 0, T Suniv < 0)
D)( Suniv < 0, G < 0, T Suniv < 0)
E)( Suniv > 0, G > 0, T Suniv > 0)

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
38
HI has a normal boiling point of -35.4°C, and its Hvap is 21.16 kJ/mol.Calculate the molar entropy of vaporization ( Svap).
A)598 J/K·mol
B)68.6 J/K·mol
C)75.2 J/K·mol
D)0.068 J/K·mol
E)89.0 J/K·mol
A)598 J/K·mol
B)68.6 J/K·mol
C)75.2 J/K·mol
D)0.068 J/K·mol
E)89.0 J/K·mol

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
39
Which of the following is consistent with a spontaneous process in the forward direction?
A)( Suniv > 0, G < 0, T Suniv > 0)
B)( Suniv < 0, G > 0, T Suniv < 0)
C)( Suniv > 0, G < 0, T Suniv < 0)
D)( Suniv < 0, G < 0, T Suniv < 0)
E)( Suniv > 0, G > 0, T Suniv > 0)
A)( Suniv > 0, G < 0, T Suniv > 0)
B)( Suniv < 0, G > 0, T Suniv < 0)
C)( Suniv > 0, G < 0, T Suniv < 0)
D)( Suniv < 0, G < 0, T Suniv < 0)
E)( Suniv > 0, G > 0, T Suniv > 0)

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
40
For the reaction H2(g)+ S(s) H2S(g), H° = -20.2 kJ/mol and S° = +43.1 J/K·mol.Which of these statements is true?
A)The reaction is only spontaneous at low temperatures.
B)The reaction is spontaneous at all temperatures.
C)( G° becomes less favorable as temperature increases.)
D)The reaction is spontaneous only at high temperatures.
E)The reaction is at equilibrium at 25°C under standard conditions.
A)The reaction is only spontaneous at low temperatures.
B)The reaction is spontaneous at all temperatures.
C)( G° becomes less favorable as temperature increases.)
D)The reaction is spontaneous only at high temperatures.
E)The reaction is at equilibrium at 25°C under standard conditions.

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
41
The equilibrium constant for the reaction AgBr(s) ![<strong>The equilibrium constant for the reaction AgBr(s) <sub> </sub> Ag<sup>+</sup>(aq)+ Br<sup>- </sup>(aq)is the solubility product constant, K<sub>sp</sub> = 7.7 × 10<sup>-13</sup> at 25°C.Calculate \Delta G for the reaction when [Ag<sup>+</sup>] = 1.0 × 10<sup>-2</sup> M and [Br<sup>-</sup>] = 1.0 × 10<sup>-3</sup> M.Is the reaction spontaneous or nonspontaneous at these concentrations?</strong> A)( \Delta G = 69.1 kJ/mol, nonspontaneous) B)( \Delta G = -69.1 kJ/mol, spontaneous) C)( \Delta G = 97.5 kJ/mol, spontaneous) D)( \Delta G = 40.6 kJ/mol, nonspontaneous) E)( \Delta G = -97.5 kJ/mol, nonspontaneous)](https://d2lvgg3v3hfg70.cloudfront.net/TB3246/11ea7cbf_8f2e_3894_a2ab_03d9e9e6e782_TB3246_11.jpg) Ag+(aq)+ Br- (aq)is the solubility product constant, Ksp = 7.7 × 10-13 at 25°C.Calculate G for the reaction when [Ag+] = 1.0 × 10-2 M and [Br-] = 1.0 × 10-3 M.Is the reaction spontaneous or nonspontaneous at these concentrations?
Ag+(aq)+ Br- (aq)is the solubility product constant, Ksp = 7.7 × 10-13 at 25°C.Calculate G for the reaction when [Ag+] = 1.0 × 10-2 M and [Br-] = 1.0 × 10-3 M.Is the reaction spontaneous or nonspontaneous at these concentrations?
A)( G = 69.1 kJ/mol, nonspontaneous)
B)( G = -69.1 kJ/mol, spontaneous)
C)( G = 97.5 kJ/mol, spontaneous)
D)( G = 40.6 kJ/mol, nonspontaneous)
E)( G = -97.5 kJ/mol, nonspontaneous)
![<strong>The equilibrium constant for the reaction AgBr(s) <sub> </sub> Ag<sup>+</sup>(aq)+ Br<sup>- </sup>(aq)is the solubility product constant, K<sub>sp</sub> = 7.7 × 10<sup>-13</sup> at 25°C.Calculate \Delta G for the reaction when [Ag<sup>+</sup>] = 1.0 × 10<sup>-2</sup> M and [Br<sup>-</sup>] = 1.0 × 10<sup>-3</sup> M.Is the reaction spontaneous or nonspontaneous at these concentrations?</strong> A)( \Delta G = 69.1 kJ/mol, nonspontaneous) B)( \Delta G = -69.1 kJ/mol, spontaneous) C)( \Delta G = 97.5 kJ/mol, spontaneous) D)( \Delta G = 40.6 kJ/mol, nonspontaneous) E)( \Delta G = -97.5 kJ/mol, nonspontaneous)](https://d2lvgg3v3hfg70.cloudfront.net/TB3246/11ea7cbf_8f2e_3894_a2ab_03d9e9e6e782_TB3246_11.jpg) Ag+(aq)+ Br- (aq)is the solubility product constant, Ksp = 7.7 × 10-13 at 25°C.Calculate G for the reaction when [Ag+] = 1.0 × 10-2 M and [Br-] = 1.0 × 10-3 M.Is the reaction spontaneous or nonspontaneous at these concentrations?
Ag+(aq)+ Br- (aq)is the solubility product constant, Ksp = 7.7 × 10-13 at 25°C.Calculate G for the reaction when [Ag+] = 1.0 × 10-2 M and [Br-] = 1.0 × 10-3 M.Is the reaction spontaneous or nonspontaneous at these concentrations?A)( G = 69.1 kJ/mol, nonspontaneous)
B)( G = -69.1 kJ/mol, spontaneous)
C)( G = 97.5 kJ/mol, spontaneous)
D)( G = 40.6 kJ/mol, nonspontaneous)
E)( G = -97.5 kJ/mol, nonspontaneous)

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
42
Calculate the equilibrium constant for the decomposition of water 2H2O(l) 
2H2(g)+ O2(g)
At 25°C, given that G°f (H2O(l))= -237.2 kJ/mol.
A)0.83
B)6.3 × 10-84
C)2.5 × 10-42
D)1.6 × 1083
E)4.7 × 105
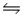
2H2(g)+ O2(g)
At 25°C, given that G°f (H2O(l))= -237.2 kJ/mol.
A)0.83
B)6.3 × 10-84
C)2.5 × 10-42
D)1.6 × 1083
E)4.7 × 105

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
43
At 1500°C the equilibrium constant for the reaction CO(g)+ 2H2(g)  CH3OH(g)has the value Kp = 1.4 × 10-7.Calculate G° for this reaction at 1500°C.
CH3OH(g)has the value Kp = 1.4 × 10-7.Calculate G° for this reaction at 1500°C.
A)105 kJ/mol
B)1.07 kJ/mol
C)-233 kJ/mol
D)-105 kJ/mol
E)233 kJ/mol
 CH3OH(g)has the value Kp = 1.4 × 10-7.Calculate G° for this reaction at 1500°C.
CH3OH(g)has the value Kp = 1.4 × 10-7.Calculate G° for this reaction at 1500°C.A)105 kJ/mol
B)1.07 kJ/mol
C)-233 kJ/mol
D)-105 kJ/mol
E)233 kJ/mol

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
44
Nitrosyl chloride (NOCl)decomposes at elevated temperatures according to the equation 2NOCl(g) 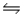
2NO(g)+ Cl2(g).Calculate Kp for this reaction at 227°C.( H° = 81.2 kJ/mol, S° = 128 J/K·mol)
A)1.59 × 10-2
B)2.10 × 10-7
C)62.8
D)4.90 × 106
E)3.20 × 109
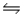
2NO(g)+ Cl2(g).Calculate Kp for this reaction at 227°C.( H° = 81.2 kJ/mol, S° = 128 J/K·mol)
A)1.59 × 10-2
B)2.10 × 10-7
C)62.8
D)4.90 × 106
E)3.20 × 109

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
45
Which of the following is consistent with a reaction that proceeds spontaneously in the forward direction?
A)( G > 0, Q < K)
B)( G° = 0, Q = K)
C)( G < 0, Q > K)
D)( G° > 0, Q = K)
E)( G < 0, Q < K)
A)( G > 0, Q < K)
B)( G° = 0, Q = K)
C)( G < 0, Q > K)
D)( G° > 0, Q = K)
E)( G < 0, Q < K)

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
46
Find the temperature at which the reaction N2O4(g) 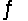 2NO2(g)will be in equilibrium when both gases are present at partial pressures of 1.00 atm.
2NO2(g)will be in equilibrium when both gases are present at partial pressures of 1.00 atm. 
A)300°C
B)28°C
C)55°C
D)32°C
E)562°C
 2NO2(g)will be in equilibrium when both gases are present at partial pressures of 1.00 atm.
2NO2(g)will be in equilibrium when both gases are present at partial pressures of 1.00 atm. 
A)300°C
B)28°C
C)55°C
D)32°C
E)562°C

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
47
Which of the following is consistent with a reaction that proceeds spontaneously in the reverse direction (assume all variables are in terms of the forward direction only)?
A)( G > 0, Q < K)
B)( G° = 0, Q = K)
C)( G < 0, Q > K)
D)( G° > 0, Q = K)
E)( G > 0, Q > K)
A)( G > 0, Q < K)
B)( G° = 0, Q = K)
C)( G < 0, Q > K)
D)( G° > 0, Q = K)
E)( G > 0, Q > K)

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
48
For the reaction 2 SO2(g)+ O2(g) 2 SO3(g), if initially P(SO2)= 1.2 atm, P(O2)= 1.8 atm, and P(SO3)= 2.1 atm, calculate G for this reaction at 25°C.The following data is valid at 25°C: 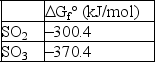
A)-140.0 kJ/mol
B)-137.6 kJ/mol
C)-138.7 kJ/mol
D)1,174.7 kJ/mol
E)-141.3 kJ/mol
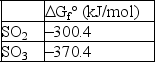
A)-140.0 kJ/mol
B)-137.6 kJ/mol
C)-138.7 kJ/mol
D)1,174.7 kJ/mol
E)-141.3 kJ/mol

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
49
The equilibrium constant at 427°C for the reaction N2(g)+ 3H2(g)  2NH3(g)is Kp = 9.4 × 10-5.Calculate the value of G° for the reaction under these conditions.
2NH3(g)is Kp = 9.4 × 10-5.Calculate the value of G° for the reaction under these conditions.
A)-33 kJ/mol
B)-54 kJ/mol
C)54 kJ/mol
D)33 kJ/mol
E)1.3 J/mol
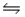 2NH3(g)is Kp = 9.4 × 10-5.Calculate the value of G° for the reaction under these conditions.
2NH3(g)is Kp = 9.4 × 10-5.Calculate the value of G° for the reaction under these conditions.A)-33 kJ/mol
B)-54 kJ/mol
C)54 kJ/mol
D)33 kJ/mol
E)1.3 J/mol

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
50
Kw for the auto-ionization of water, H2O(l) H+(aq)+ OH- (aq), is 1.0 × 10-14.What are the signs (+/-)of S° and H° for the reaction at 25°C?
A)( S° = (+)and H° = (+))
B)( S° = (+)and H° = (-))
C)( S° = (-)and H° = (+))
D)( S° = (-)and H° = (-))
A)( S° = (+)and H° = (+))
B)( S° = (+)and H° = (-))
C)( S° = (-)and H° = (+))
D)( S° = (-)and H° = (-))

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
51
Calculate G° for the combustion of ethanol vapor, C2H5OH(g), at 750°C in oxygen to form carbon dioxide and water vapor.The following data is valid at 25°C: 
A)-1407 kJ/mol
B)-2151 kJ/mol
C)-1307 kJ/mol
D)-4486 kJ/mol
E)-1377 kJ/mol

A)-1407 kJ/mol
B)-2151 kJ/mol
C)-1307 kJ/mol
D)-4486 kJ/mol
E)-1377 kJ/mol

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
52
Calculate Kp at 298 K for the reaction SO2(g)+ NO2(g) 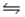 SO3(g)+ NO(g).
SO3(g)+ NO(g). 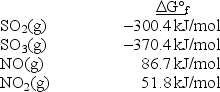
A)6.99 × 10-7
B)5.71 × 10-8
C)14.2
D)475
E)1.42 × 106
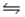 SO3(g)+ NO(g).
SO3(g)+ NO(g). 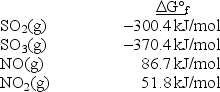
A)6.99 × 10-7
B)5.71 × 10-8
C)14.2
D)475
E)1.42 × 106

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
53
The reaction rates of many spontaneous reactions are actually very slow.Which of these statements is the best explanation for this observation?
A)(Kp for the reaction is less than one.)
B)The activation energy of the reaction is large.
C)( G° for the reaction is positive.)
D)Such reactions are endothermic.
E)The entropy change is negative.
A)(Kp for the reaction is less than one.)
B)The activation energy of the reaction is large.
C)( G° for the reaction is positive.)
D)Such reactions are endothermic.
E)The entropy change is negative.

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
54
Which of the following is consistent with a reaction at equilibrium?
A)( G = 0, Q = K)
B)( G° = 0, Q > K)
C)( G > 0, Q = K)
D)( G < 0, Q = K)
E)( G° = 0, Q < K)
A)( G = 0, Q = K)
B)( G° = 0, Q > K)
C)( G > 0, Q = K)
D)( G < 0, Q = K)
E)( G° = 0, Q < K)

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
55
The solubility product constant at 25°C for AgI(s)in water has the value 8.3 × 10-17.Calculate Grxn at 25°C for the process AgI(s) ![<strong>The solubility product constant at 25°C for AgI(s)in water has the value 8.3 × 10<sup>-17</sup>.Calculate \Delta G<sub>rxn</sub> at 25°C for the process AgI(s) <sub> </sub> Ag<sup>+</sup>(aq)+ I<sup>- </sup>(aq)where [Ag<sup>+</sup>] = 9.1 × 10<sup>-9</sup> and [I<sup>-</sup>] = 9.1 × 10<sup>-9</sup>.</strong> A)+4.4 kJ/mol B)+91.7 kJ/mol C)0.0 kJ/mol D)-91.7 kJ/mol E)-4.4 kJ/mol](https://d2lvgg3v3hfg70.cloudfront.net/TB3246/11ea7cbf_8f2e_86b8_a2ab_b1334d8022b1_TB3246_11.jpg) Ag+(aq)+ I- (aq)where [Ag+] = 9.1 × 10-9 and [I-] = 9.1 × 10-9.
Ag+(aq)+ I- (aq)where [Ag+] = 9.1 × 10-9 and [I-] = 9.1 × 10-9.
A)+4.4 kJ/mol
B)+91.7 kJ/mol
C)0.0 kJ/mol
D)-91.7 kJ/mol
E)-4.4 kJ/mol
![<strong>The solubility product constant at 25°C for AgI(s)in water has the value 8.3 × 10<sup>-17</sup>.Calculate \Delta G<sub>rxn</sub> at 25°C for the process AgI(s) <sub> </sub> Ag<sup>+</sup>(aq)+ I<sup>- </sup>(aq)where [Ag<sup>+</sup>] = 9.1 × 10<sup>-9</sup> and [I<sup>-</sup>] = 9.1 × 10<sup>-9</sup>.</strong> A)+4.4 kJ/mol B)+91.7 kJ/mol C)0.0 kJ/mol D)-91.7 kJ/mol E)-4.4 kJ/mol](https://d2lvgg3v3hfg70.cloudfront.net/TB3246/11ea7cbf_8f2e_86b8_a2ab_b1334d8022b1_TB3246_11.jpg) Ag+(aq)+ I- (aq)where [Ag+] = 9.1 × 10-9 and [I-] = 9.1 × 10-9.
Ag+(aq)+ I- (aq)where [Ag+] = 9.1 × 10-9 and [I-] = 9.1 × 10-9.A)+4.4 kJ/mol
B)+91.7 kJ/mol
C)0.0 kJ/mol
D)-91.7 kJ/mol
E)-4.4 kJ/mol

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
56
Determine the equilibrium constant Kp at 25°C for the reaction N2(g)+ 3H2(g) ![<strong>Determine the equilibrium constant K<sub>p</sub> at 25°C for the reaction N<sub>2</sub>(g)+ 3H<sub>2</sub>(g) <sub> </sub> 2NH<sub>3</sub>(g) [ \Delta G°<sub>f</sub> (NH<sub>3</sub>(g))= -16.6 kJ/mol].</strong> A)1.52 × 10<sup>-6</sup> B)6.60 × 10<sup>5</sup> C)8.28 × 10<sup>-2</sup> D)2.60 E)13.4](https://d2lvgg3v3hfg70.cloudfront.net/TB3246/11ea7cbf_8f2e_1181_a2ab_b16fc065d489_TB3246_11.jpg) 2NH3(g)
2NH3(g)
[ G°f (NH3(g))= -16.6 kJ/mol].
A)1.52 × 10-6
B)6.60 × 105
C)8.28 × 10-2
D)2.60
E)13.4
![<strong>Determine the equilibrium constant K<sub>p</sub> at 25°C for the reaction N<sub>2</sub>(g)+ 3H<sub>2</sub>(g) <sub> </sub> 2NH<sub>3</sub>(g) [ \Delta G°<sub>f</sub> (NH<sub>3</sub>(g))= -16.6 kJ/mol].</strong> A)1.52 × 10<sup>-6</sup> B)6.60 × 10<sup>5</sup> C)8.28 × 10<sup>-2</sup> D)2.60 E)13.4](https://d2lvgg3v3hfg70.cloudfront.net/TB3246/11ea7cbf_8f2e_1181_a2ab_b16fc065d489_TB3246_11.jpg) 2NH3(g)
2NH3(g)[ G°f (NH3(g))= -16.6 kJ/mol].
A)1.52 × 10-6
B)6.60 × 105
C)8.28 × 10-2
D)2.60
E)13.4

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
57
For the reaction 2C(graphite)+ H2(g) C2H2(g), G°= +209.2 kJ/mol at 25°C.If P(H2)= 100.atm, and P(C2H2)= 0.10 atm, calculate G for this reaction.
A)+207.8 kJ/mol
B)+226.3 kJ/mol
C)+192.1 kJ/mol
D)+17.3 kJ/mol
E)-16.9 kJ/mol
A)+207.8 kJ/mol
B)+226.3 kJ/mol
C)+192.1 kJ/mol
D)+17.3 kJ/mol
E)-16.9 kJ/mol

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
58
For the reaction 2NO(g)+ O2(g) 2NO2(g)if initially P(NO)= 1.5 atm, P(O2)= 1.4 atm, and P(NO2)= 2.0 atm, calculate G for this reaction at 25°C.The following data is valid at 25°C: 
A)-69.9 kJ/mol
B)-69.2 kJ/mol
C)522.1 kJ/mol
D)-79.9 kJ/mol
E)-35.0 kJ/mol

A)-69.9 kJ/mol
B)-69.2 kJ/mol
C)522.1 kJ/mol
D)-79.9 kJ/mol
E)-35.0 kJ/mol

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
59
Determine the equilibrium constant (Kp)at 25°C for the reaction

A)2.9 × 10-60
B)1.0 × 10-4
C)1.2
D)1.0 × 105
E)3.4 × 1059

A)2.9 × 10-60
B)1.0 × 10-4
C)1.2
D)1.0 × 105
E)3.4 × 1059

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
60
Predict the normal boiling point of triethylborane (C6H15B)using the following data: 
A)92°C
B)-21°C
C)21°C
D)365°C
E)256°C

A)92°C
B)-21°C
C)21°C
D)365°C
E)256°C

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
61
For the reaction CuS(s)+ H2(g) 
H2S(g)+ Cu(s),
G°f (CuS)= -53.6 kJ/mol
G°f (H2S)= -33.6 kJ/mol
H°f (CuS)= -53.1 kJ/mol
H°f (H2S)= -20.6 kJ/mol
Calculate the value of the equilibrium constant (Kp)at 798 K and 1 atm pressure.
H2S(g)+ Cu(s),
G°f (CuS)= -53.6 kJ/mol
G°f (H2S)= -33.6 kJ/mol
H°f (CuS)= -53.1 kJ/mol
H°f (H2S)= -20.6 kJ/mol
Calculate the value of the equilibrium constant (Kp)at 798 K and 1 atm pressure.

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
62
Using the thermodynamic data provided below, calculate the standard change in entropy when one mole of sodium sulfate is dissolved in water? 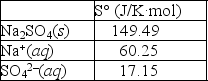 Will the solubility of sodium nitrate increase or decrease if the temperature of the system is increased?
Will the solubility of sodium nitrate increase or decrease if the temperature of the system is increased?
 Will the solubility of sodium nitrate increase or decrease if the temperature of the system is increased?
Will the solubility of sodium nitrate increase or decrease if the temperature of the system is increased?
Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
63
For the reaction SbCl5(g) 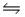
SbCl3(g)+ Cl2(g),
G°f (SbCl5)= -334.34 kJ/mol
G°f (SbCl3)= -301.25 kJ/mol
H°f (SbCl5)= -394.34 kJ/mol
H°f (SbCl3)= -313.80 kJ/mol
Calculate G at 800 K and 1 atm pressure (assume S° and H° do not change with temperature).
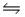
SbCl3(g)+ Cl2(g),
G°f (SbCl5)= -334.34 kJ/mol
G°f (SbCl3)= -301.25 kJ/mol
H°f (SbCl5)= -394.34 kJ/mol
H°f (SbCl3)= -313.80 kJ/mol
Calculate G at 800 K and 1 atm pressure (assume S° and H° do not change with temperature).

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
64
For the reaction CuS(s)+ H2(g) 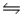
H2S(g)+ Cu(s),
G°f (CuS)= -53.6 kJ/mol
G°f (H2S)= -33.6 kJ/mol
H°f (CuS)= -53.1 kJ/mol
H°f (H2S)= - 20.6 kJ/mol
Will this reaction proceed spontaneously at 298 K and 1 atm pressure?
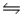
H2S(g)+ Cu(s),
G°f (CuS)= -53.6 kJ/mol
G°f (H2S)= -33.6 kJ/mol
H°f (CuS)= -53.1 kJ/mol
H°f (H2S)= - 20.6 kJ/mol
Will this reaction proceed spontaneously at 298 K and 1 atm pressure?

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
65
For the reaction SbCl5(g) 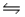
SbCl3(g)+ Cl2(g),
G°f (SbCl5)= -334.34 kJ/mol
G°f (SbCl3)= -301.25 kJ/mol
H°f (SbCl5)= -394.34 kJ/mol
H°f (SbCl3)= -313.80 kJ/mol
Calculate the value of the equilibrium constant (Kp)for this reaction at 298 K.
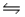
SbCl3(g)+ Cl2(g),
G°f (SbCl5)= -334.34 kJ/mol
G°f (SbCl3)= -301.25 kJ/mol
H°f (SbCl5)= -394.34 kJ/mol
H°f (SbCl3)= -313.80 kJ/mol
Calculate the value of the equilibrium constant (Kp)for this reaction at 298 K.

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
66
The standard free energy of formation of gaseous hydrogen iodide is 1.30 kJ/mol at 25°C.Find Kp for the reaction H2(g)+ I2(s) 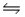 2HI(g)at this temperature.
2HI(g)at this temperature.
A)7.0
B)7100
C)1.0
D)2.4
E)2.9
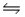 2HI(g)at this temperature.
2HI(g)at this temperature.A)7.0
B)7100
C)1.0
D)2.4
E)2.9

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
67
In the gas phase, formic acid forms a dimmer, 2HCOOH(g) 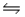 (HCOOH)2(g).For this reaction, H° = -60.1 kJ/mol and G° = -13.9 kJ/mol at 25°C.Find the equilibrium constant (Kp)for this reaction at 75 °C.
(HCOOH)2(g).For this reaction, H° = -60.1 kJ/mol and G° = -13.9 kJ/mol at 25°C.Find the equilibrium constant (Kp)for this reaction at 75 °C.
A)8960
B)273
C)0.120
D)8.33
E)1.12 × 10-4
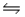 (HCOOH)2(g).For this reaction, H° = -60.1 kJ/mol and G° = -13.9 kJ/mol at 25°C.Find the equilibrium constant (Kp)for this reaction at 75 °C.
(HCOOH)2(g).For this reaction, H° = -60.1 kJ/mol and G° = -13.9 kJ/mol at 25°C.Find the equilibrium constant (Kp)for this reaction at 75 °C.A)8960
B)273
C)0.120
D)8.33
E)1.12 × 10-4

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
68
For the reaction HCONH2(g) 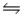 NH3(g)+ CO(g), Kc = 4.84 at 400 K.If H° for this reaction is 29 kJ/mol, find Kc at 500 K.
NH3(g)+ CO(g), Kc = 4.84 at 400 K.If H° for this reaction is 29 kJ/mol, find Kc at 500 K.
A)5.8
B)0.17
C)27
D)0.88
E)10.3
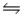 NH3(g)+ CO(g), Kc = 4.84 at 400 K.If H° for this reaction is 29 kJ/mol, find Kc at 500 K.
NH3(g)+ CO(g), Kc = 4.84 at 400 K.If H° for this reaction is 29 kJ/mol, find Kc at 500 K.A)5.8
B)0.17
C)27
D)0.88
E)10.3

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
69
Using the thermodynamic data provided below, calculate the standard change in entropy when one mole of sodium nitrate is dissolved in water? 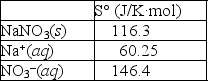 Will the solubility of sodium nitrate increase or decrease if the temperature of the system is increased?
Will the solubility of sodium nitrate increase or decrease if the temperature of the system is increased?
 Will the solubility of sodium nitrate increase or decrease if the temperature of the system is increased?
Will the solubility of sodium nitrate increase or decrease if the temperature of the system is increased?
Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
70
The free energy of formation of nitric oxide, NO, at 1000 K (roughly the temperature in an automobile engine during ignition)is about 78 kJ/mol.Calculate the equilibrium constant Kp for the reaction N2(g)+ O2(g) 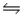
2NO(g)at this temperature.
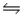
2NO(g)at this temperature.

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
71
Assuming S° and H° do not vary with temperature, at what temperature will the reaction shown below become spontaneous?
C(s)+ H2O(g) H2(g)+ CO(s)( S° = 133.6 J/K·mol; H° = 131.3 kJ/mol)
C(s)+ H2O(g) H2(g)+ CO(s)( S° = 133.6 J/K·mol; H° = 131.3 kJ/mol)

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
72
For the reaction SbCl5(g) 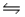
SbCl3(g)+ Cl2(g),
G°f (SbCl5)= -334.34 kJ/mol
G°f (SbCl3)= -301.25 kJ/mol
H°f (SbCl5)= -394.34 kJ/mol
H°f (SbCl3)= -313.80 kJ/mol
Calculate the value of the equilibrium constant (Kp)at 800 K and 1 atm pressure.
SbCl3(g)+ Cl2(g),
G°f (SbCl5)= -334.34 kJ/mol
G°f (SbCl3)= -301.25 kJ/mol
H°f (SbCl5)= -394.34 kJ/mol
H°f (SbCl3)= -313.80 kJ/mol
Calculate the value of the equilibrium constant (Kp)at 800 K and 1 atm pressure.

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
73
Find the temperature at which Kp = 42.0 for the reaction H2(g)+ I2(g) ![<strong>Find the temperature at which K<sub>p</sub> = 42.0 for the reaction H<sub>2</sub>(g)+ I<sub>2</sub>(g) <sub> </sub> 2HI(g).[Given: at 25°C, for H<sub>2</sub>(g), \Delta H°<sub>f</sub> = 0, S° = 131.0 J/mol·K; for I<sub>2</sub>(g), \Delta H°<sub>f</sub> = 62.26 kJ/mol, S° = 260.6 J/mol·K; for HI(g), \Delta H°<sub>f</sub> = 25.9 kJ/mol, S° = 206.3 J/mol·K; assume that \Delta H° and \Delta S° are independent of temperature.]</strong> A)1040 K B)168 K C)539 K D)1400 K E)34,200 K](https://d2lvgg3v3hfg70.cloudfront.net/TB3246/11ea7cbf_8f2e_fbf0_a2ab_4dbaa96e7fd3_TB3246_11.jpg) 2HI(g).[Given: at 25°C, for H2(g), H°f = 0, S° = 131.0 J/mol·K; for I2(g), H°f = 62.26 kJ/mol, S° = 260.6 J/mol·K; for HI(g), H°f = 25.9 kJ/mol, S° = 206.3 J/mol·K; assume that H° and S° are independent of temperature.]
2HI(g).[Given: at 25°C, for H2(g), H°f = 0, S° = 131.0 J/mol·K; for I2(g), H°f = 62.26 kJ/mol, S° = 260.6 J/mol·K; for HI(g), H°f = 25.9 kJ/mol, S° = 206.3 J/mol·K; assume that H° and S° are independent of temperature.]
A)1040 K
B)168 K
C)539 K
D)1400 K
E)34,200 K
![<strong>Find the temperature at which K<sub>p</sub> = 42.0 for the reaction H<sub>2</sub>(g)+ I<sub>2</sub>(g) <sub> </sub> 2HI(g).[Given: at 25°C, for H<sub>2</sub>(g), \Delta H°<sub>f</sub> = 0, S° = 131.0 J/mol·K; for I<sub>2</sub>(g), \Delta H°<sub>f</sub> = 62.26 kJ/mol, S° = 260.6 J/mol·K; for HI(g), \Delta H°<sub>f</sub> = 25.9 kJ/mol, S° = 206.3 J/mol·K; assume that \Delta H° and \Delta S° are independent of temperature.]</strong> A)1040 K B)168 K C)539 K D)1400 K E)34,200 K](https://d2lvgg3v3hfg70.cloudfront.net/TB3246/11ea7cbf_8f2e_fbf0_a2ab_4dbaa96e7fd3_TB3246_11.jpg) 2HI(g).[Given: at 25°C, for H2(g), H°f = 0, S° = 131.0 J/mol·K; for I2(g), H°f = 62.26 kJ/mol, S° = 260.6 J/mol·K; for HI(g), H°f = 25.9 kJ/mol, S° = 206.3 J/mol·K; assume that H° and S° are independent of temperature.]
2HI(g).[Given: at 25°C, for H2(g), H°f = 0, S° = 131.0 J/mol·K; for I2(g), H°f = 62.26 kJ/mol, S° = 260.6 J/mol·K; for HI(g), H°f = 25.9 kJ/mol, S° = 206.3 J/mol·K; assume that H° and S° are independent of temperature.]A)1040 K
B)168 K
C)539 K
D)1400 K
E)34,200 K

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
74
Find the temperature at which Kp = 4.00 for the reaction N2O4(g) ![<strong>Find the temperature at which K<sub>p</sub> = 4.00 for the reaction N<sub>2</sub>O<sub>4</sub>(g) <sub> </sub> 2NO<sub>2</sub>(g).[Given: at 25°C, for NO<sub>2</sub>(g), \Delta H°<sub>f</sub> = 33.85 kJ/mol, S° = 240.46 J/mol·K; for N<sub>2</sub>O<sub>4</sub>(g), \Delta H°<sub>f</sub> = 9.66 kJ/mol, S° = 304.3 J/mol·K; assume that \Delta H° and \Delta S° are independent of temperature.]</strong> A)197 °C B)56 °C C)36 °C D)79 °C E)476°C](https://d2lvgg3v3hfg70.cloudfront.net/TB3246/11ea7cbf_8f2e_fbef_a2ab_7d28e48ccb81_TB3246_11.jpg) 2NO2(g).[Given: at 25°C, for NO2(g), H°f = 33.85 kJ/mol, S° = 240.46 J/mol·K; for N2O4(g), H°f = 9.66 kJ/mol, S° = 304.3 J/mol·K; assume that H° and S° are independent of temperature.]
2NO2(g).[Given: at 25°C, for NO2(g), H°f = 33.85 kJ/mol, S° = 240.46 J/mol·K; for N2O4(g), H°f = 9.66 kJ/mol, S° = 304.3 J/mol·K; assume that H° and S° are independent of temperature.]
A)197 °C
B)56 °C
C)36 °C
D)79 °C
E)476°C
![<strong>Find the temperature at which K<sub>p</sub> = 4.00 for the reaction N<sub>2</sub>O<sub>4</sub>(g) <sub> </sub> 2NO<sub>2</sub>(g).[Given: at 25°C, for NO<sub>2</sub>(g), \Delta H°<sub>f</sub> = 33.85 kJ/mol, S° = 240.46 J/mol·K; for N<sub>2</sub>O<sub>4</sub>(g), \Delta H°<sub>f</sub> = 9.66 kJ/mol, S° = 304.3 J/mol·K; assume that \Delta H° and \Delta S° are independent of temperature.]</strong> A)197 °C B)56 °C C)36 °C D)79 °C E)476°C](https://d2lvgg3v3hfg70.cloudfront.net/TB3246/11ea7cbf_8f2e_fbef_a2ab_7d28e48ccb81_TB3246_11.jpg) 2NO2(g).[Given: at 25°C, for NO2(g), H°f = 33.85 kJ/mol, S° = 240.46 J/mol·K; for N2O4(g), H°f = 9.66 kJ/mol, S° = 304.3 J/mol·K; assume that H° and S° are independent of temperature.]
2NO2(g).[Given: at 25°C, for NO2(g), H°f = 33.85 kJ/mol, S° = 240.46 J/mol·K; for N2O4(g), H°f = 9.66 kJ/mol, S° = 304.3 J/mol·K; assume that H° and S° are independent of temperature.]A)197 °C
B)56 °C
C)36 °C
D)79 °C
E)476°C

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
75
A sample of solid naphthalene is introduced into an evacuated flask.Use the data below to calculate the equilibrium vapor pressure of naphthalene (C10H8)in the flask at 35°C. 
A)890.mmHg
B)0.21 mmHg
C)696 mmHg
D)0.086 mmHg
E)833 mmHg

A)890.mmHg
B)0.21 mmHg
C)696 mmHg
D)0.086 mmHg
E)833 mmHg

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
76
For the reaction CuS(s)+ H2(g) 
H2S(g)+ Cu(s),
G°f (CuS)= -53.6 kJ/mol
G°f (H2S)= -33.6 kJ/mol
H°f (CuS)= -53.1 kJ/mol
H°f (H2S)= - 20.6 kJ/mol
Calculate the value of the equilibrium constant (Kp)for this reaction at 298 K.
H2S(g)+ Cu(s),
G°f (CuS)= -53.6 kJ/mol
G°f (H2S)= -33.6 kJ/mol
H°f (CuS)= -53.1 kJ/mol
H°f (H2S)= - 20.6 kJ/mol
Calculate the value of the equilibrium constant (Kp)for this reaction at 298 K.

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
77
In the gas phase, methyl isocyanate (CH3NC)isomerizes to acetonitrile (CH3CN), H3C-N C (g) 
H3C-C N (g)
With H° = -89.5 kJ/mol and G° = - 73.8 kJ/mol at 25°C.Find the equilibrium constant for this reaction at 100°C.
A)1.68 × 10-10
B)5.96 × 109
C)2.16 × 1010
D)4.63 × 10-11
E)8.64 × 1012
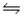
H3C-C N (g)
With H° = -89.5 kJ/mol and G° = - 73.8 kJ/mol at 25°C.Find the equilibrium constant for this reaction at 100°C.
A)1.68 × 10-10
B)5.96 × 109
C)2.16 × 1010
D)4.63 × 10-11
E)8.64 × 1012

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
78
For the reaction SbCl5(g) 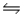
SbCl3(g)+ Cl2(g),
G°f (SbCl5)= -334.34 kJ/mol
G°f (SbCl3)= -301.25 kJ/mol
H°f (SbCl5)= -394.34 kJ/mol
H°f (SbCl3)= -313.80 kJ/mol
Will this reaction proceed spontaneously at 298 K and 1 atm pressure?
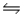
SbCl3(g)+ Cl2(g),
G°f (SbCl5)= -334.34 kJ/mol
G°f (SbCl3)= -301.25 kJ/mol
H°f (SbCl5)= -394.34 kJ/mol
H°f (SbCl3)= -313.80 kJ/mol
Will this reaction proceed spontaneously at 298 K and 1 atm pressure?

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
79
Rubidium has a heat of vaporization of 69.0 kJ/mol at its boiling point (686°C).Calculate S for this process, Rb(l) Rb(g), at 1 atm and 686°C.

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck
80
For the reaction CuS(s)+ H2(g) 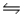
H2S(g)+ Cu(s),
G°f (CuS)= -53.6 kJ/mol
G°f (H2S)= -33.6 kJ/mol
H°f (CuS)= -53.1 kJ/mol
H°f (H2S)= -20.6 kJ/mol
Calculate G at 798 K and 1 atm pressure (assume S° and H° do not change with temperature).
H2S(g)+ Cu(s),
G°f (CuS)= -53.6 kJ/mol
G°f (H2S)= -33.6 kJ/mol
H°f (CuS)= -53.1 kJ/mol
H°f (H2S)= -20.6 kJ/mol
Calculate G at 798 K and 1 atm pressure (assume S° and H° do not change with temperature).

Unlock Deck
Unlock for access to all 128 flashcards in this deck.
Unlock Deck
k this deck



