Deck 16: Coordination Compounds-The Colorful Chemistry of Metals
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question

Unlock Deck
Sign up to unlock the cards in this deck!
Unlock Deck
Unlock Deck
1/153
Play
Full screen (f)
Deck 16: Coordination Compounds-The Colorful Chemistry of Metals
1
Which of the following species is a Lewis base?
A)H+
B)Cs+
C)NF3
D)NF4+
E)NH4+
A)H+
B)Cs+
C)NF3
D)NF4+
E)NH4+
NF3
2
A Lewis base is __________
A)an electron-pair acceptor.
B)an electron-pair donor.
C)a proton donor.
D)a proton acceptor.
E)never viewed also as a Brønsted-Lowry base.
A)an electron-pair acceptor.
B)an electron-pair donor.
C)a proton donor.
D)a proton acceptor.
E)never viewed also as a Brønsted-Lowry base.
an electron-pair donor.
3
Prussian blue has an idealized chemical formula of Fe4[Fe(CN)6]3.What is the charge on the counter ions? (Note: the charge on the iron ion in the complex ion is an integer.)
A)+6
B)+5
C)+4
D)+3
E)+2
A)+6
B)+5
C)+4
D)+3
E)+2
+3
4
What is the oxidation state of cobalt in the [Co(NH3)4Cl2]+ complex ion?
A)+1
B)+2
C)+3
D)+4
E)+6
A)+1
B)+2
C)+3
D)+4
E)+6

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
5
What is the oxidation state of the metal atom in the complex ion in K[Pt(NH3)Cl5]?
A)+6
B)+5
C)+4
D)+3
E)+2
A)+6
B)+5
C)+4
D)+3
E)+2

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
6
Identify the Lewis base in the following reaction: SO2(g)+ H2O(  ) H2SO3(aq)
) H2SO3(aq)
A)SO2
B)H2O
C)H2SO3
D)None of these is a base.
E)All of these are bases.
 ) H2SO3(aq)
) H2SO3(aq)A)SO2
B)H2O
C)H2SO3
D)None of these is a base.
E)All of these are bases.

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
7
In the following reaction,which species is the Lewis base? Cu2+(aq)+ NH3(aq) Cu(NH3)2+(aq)
A)Cu2+
B)NH3
C)[Cu(NH3)]2+
D)None of these is a base.
E)All of these are bases.
A)Cu2+
B)NH3
C)[Cu(NH3)]2+
D)None of these is a base.
E)All of these are bases.

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
8
Identify the Lewis acid in the following reaction: PH3(aq)+ H+(aq) PH4+(aq)
A)PH4+
B)H+
C)PH3
D)None of these is an acid.
E)All of these are acids.
A)PH4+
B)H+
C)PH3
D)None of these is an acid.
E)All of these are acids.

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
9
When NF3 reacts with BF3 to form F3NBF3,__________
A)a complex ion is formed.
B)an ionic bond is formed.
C)the bond is formed by sharing an electron from NF3 and an electron from BF3.
D)a coordinate covalent bond is formed.
E)a covalent bond is formed.
A)a complex ion is formed.
B)an ionic bond is formed.
C)the bond is formed by sharing an electron from NF3 and an electron from BF3.
D)a coordinate covalent bond is formed.
E)a covalent bond is formed.

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
10
A ligand is any __________ forming a coordinate bond to a metal cation.
A)Lewis acid
B)ion
C)Lewis base
D)organic compound
E)species
A)Lewis acid
B)ion
C)Lewis base
D)organic compound
E)species

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
11
A Lewis acid is __________
A)a proton donor.
B)a proton acceptor.
C)an electron-pair donor.
D)an electron-pair acceptor.
E)is never viewed also as a Brønsted-Lowry acid.
A)a proton donor.
B)a proton acceptor.
C)an electron-pair donor.
D)an electron-pair acceptor.
E)is never viewed also as a Brønsted-Lowry acid.

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
12
A Lewis acid is any species capable of __________ an electron pair.
A)accepting
B)donating
C)creating
D)neutralizing
E)losing
A)accepting
B)donating
C)creating
D)neutralizing
E)losing

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
13
Identify the Lewis base in the following reaction: PH3(aq)+ H+(aq) PH4+(aq)
A)PH4+
B)H+
C)PH3
D)None of these is a base.
E)All of these are bases.
A)PH4+
B)H+
C)PH3
D)None of these is a base.
E)All of these are bases.

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
14
Identify the Lewis acid in the following reaction: NH3(aq)+ H2O(  ) NH4+(aq)+ OH-(aq)
) NH4+(aq)+ OH-(aq)
A)NH3
B)H2O
C)NH4+
D)OH-
E)None of these is a base.
 ) NH4+(aq)+ OH-(aq)
) NH4+(aq)+ OH-(aq)A)NH3
B)H2O
C)NH4+
D)OH-
E)None of these is a base.

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
15
Prussian blue has an idealized chemical formula of Fe4[Fe(CN)6]3.What is the charge on the iron atom in the complex ion? (Note: the charge on the iron counter ions is an integer.)
A)+6
B)+5
C)+4
D)+3
E)+2
A)+6
B)+5
C)+4
D)+3
E)+2

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
16
A Lewis base is any species capable of __________ an electron pair.
A)accepting
B)donating
C)creating
D)neutralizing
E)gaining
A)accepting
B)donating
C)creating
D)neutralizing
E)gaining

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
17
A coordinate covalent bond forms when a chemical species __________
A)donates one electron to another species to form a covalent bond.
B)accepts a pair of electrons and becomes an anion.
C)accepts one electron from another species to form a covalent bond.
D)accepts an electron and becomes an anion.
E)donates a pair of electrons to another species to form a covalent bond.
A)donates one electron to another species to form a covalent bond.
B)accepts a pair of electrons and becomes an anion.
C)accepts one electron from another species to form a covalent bond.
D)accepts an electron and becomes an anion.
E)donates a pair of electrons to another species to form a covalent bond.

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
18
Which of the following statements regarding complex ions and coordination compounds is NOT correct?
A)Ligands function as electron pair donors.
B)A metal cation acts as a Lewis acid in a complex ion.
C)Ligands occupy the inner coordination sphere of the cation.
D)The coordination number refers to the number of electron pairs surrounding a metal ion in a complex.
E)Counter ions form coordinate covalent bonds with the ligands in a complex.
A)Ligands function as electron pair donors.
B)A metal cation acts as a Lewis acid in a complex ion.
C)Ligands occupy the inner coordination sphere of the cation.
D)The coordination number refers to the number of electron pairs surrounding a metal ion in a complex.
E)Counter ions form coordinate covalent bonds with the ligands in a complex.

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
19
In the following reaction,which species is the Lewis acid? Cu2+(aq)+ NH3(aq) Cu(NH3)2+(aq)
A)Cu2+
B)NH3
C)[Cu(NH3)]2+
D)None of these is an acid.
E)All of these are acids.
A)Cu2+
B)NH3
C)[Cu(NH3)]2+
D)None of these is an acid.
E)All of these are acids.

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
20
What is the oxidation state of nickel in tetracarbonylnickel,Ni(CO)4?
A)0
B)+2
C)+4
D)+6
E)+8
A)0
B)+2
C)+4
D)+6
E)+8

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
21
The correct name for the complex ion [Co(NH3)5Cl]2+ is __________
A)pentamminechlorocobaltate(III).
B)pentamminechlorocobalt(II).
C)chloropentamminecobalt(III).
D)pentamminechlorocobalt(III).
E)pentamminechlorocobalt.
A)pentamminechlorocobaltate(III).
B)pentamminechlorocobalt(II).
C)chloropentamminecobalt(III).
D)pentamminechlorocobalt(III).
E)pentamminechlorocobalt.

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
22
What is the coordination number around the Co ion in [Co(NH3)6][CrF6]?
A)2
B)1
C)6
D)5
E)7
A)2
B)1
C)6
D)5
E)7

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
23
What is the coordination number around the Cr ion in [Co(NH3)6][CrF6]?
A)2
B)1
C)6
D)5
E)7
A)2
B)1
C)6
D)5
E)7

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
24
What is the coordination number of cobalt in the [Co(NH3)4Cl2]+ complex ion?
A)2
B)3
C)4
D)4.0
E)6
A)2
B)3
C)4
D)4.0
E)6

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
25
The correct name for the compound [Fe(NH3)5H2O](NO3)2 is __________
A)pentammineaquairon(II)nitrate.
B)pentammineaquairon(III)nitrate.
C)pentammineaquaferrate(II)nitrate.
D)aquapentammineiron(II)nitrate.
E)pentamineaquadinitroiron(III).
A)pentammineaquairon(II)nitrate.
B)pentammineaquairon(III)nitrate.
C)pentammineaquaferrate(II)nitrate.
D)aquapentammineiron(II)nitrate.
E)pentamineaquadinitroiron(III).

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
26
For the coordination compound K3[ZrF7],identify,in order,the charge on the complex ion,the oxidation state of the metal,and the coordination number of the metal.
A)-3,+4,7
B)-3,+7,3
C)+3,-4,7
D)+4,-7,3
E)None of these choices is correct.
A)-3,+4,7
B)-3,+7,3
C)+3,-4,7
D)+4,-7,3
E)None of these choices is correct.

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
27
What is the coordination number of the metal atom in the complex ion in K[Pt(NH3)Cl5]?
A)7
B)6
C)5
D)4
E)3
A)7
B)6
C)5
D)4
E)3

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
28
For the coordination compound [Zn(NH3)4]Cl2,identify,in order,the charge on the complex ion,the coordination number of the metal,and the number of counter ions present.
A)-2,4,2
B)+2,4,4
C)+4,6,0
D)+4,4,2
E)None of these choices is correct.
A)-2,4,2
B)+2,4,4
C)+4,6,0
D)+4,4,2
E)None of these choices is correct.

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
29
How many ions are formed when [Co(NH3)5Cl]Cl2 dissolves in water?
A)1
B)2
C)3
D)4
E)5
A)1
B)2
C)3
D)4
E)5

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
30
What is the coordination number for copper in the compound K2[CuCl4]?
A)6
B)4
C)3
D)2
E)5
A)6
B)4
C)3
D)2
E)5

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
31
For the coordination compound K3[Fe(CN)6],identify,in order,the charge on the complex ion,the oxidation state of the metal,and the coordination number of the metal.
A)3+,-3,6
B)0,0,6
C)-3,+3,3
D)-3,+3,6
E)None of these choices is correct.
A)3+,-3,6
B)0,0,6
C)-3,+3,3
D)-3,+3,6
E)None of these choices is correct.

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
32
What is the chemical formula of aminetrichloroplatinate(II)?
A)[Pt2(NH3)Cl3]-
B)[Pt(NH3)3Cl]-
C)[Pt(NH3)Cl3]2-
D)[Pt(NH3)Cl3]+
E)[Pt(NH3)Cl3]-
A)[Pt2(NH3)Cl3]-
B)[Pt(NH3)3Cl]-
C)[Pt(NH3)Cl3]2-
D)[Pt(NH3)Cl3]+
E)[Pt(NH3)Cl3]-

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
33
The correct name for K2[CuCl4] is __________
A)potassium copper chloride.
B)potassium tetrachlorocuprate(II).
C)dipotassium chlorocuprate(II).
D)potassium tetrachlorocopper(II).
E)dipotassium tetrachlorocopper(II).
A)potassium copper chloride.
B)potassium tetrachlorocuprate(II).
C)dipotassium chlorocuprate(II).
D)potassium tetrachlorocopper(II).
E)dipotassium tetrachlorocopper(II).

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
34
What is the coordination number for [Pt(NH3)4]Cl2?
A)2
B)3
C)6
D)4
E)5
A)2
B)3
C)6
D)4
E)5

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
35
Which of the following releases the most ions when dissolved in water?
A)[Co(NH3)4Cl2](NO3)2
B)[Ni(NH3)5(H2O)]Cl2
C)Pt(NH3)3Cl4
D)K3[Fe(C2O4)3]
E)Na4[Fe(CN)6]
A)[Co(NH3)4Cl2](NO3)2
B)[Ni(NH3)5(H2O)]Cl2
C)Pt(NH3)3Cl4
D)K3[Fe(C2O4)3]
E)Na4[Fe(CN)6]

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
36
The correct name for [Co(NH3)5Cl]Cl2 is __________
A)chloropentamminecobalt(III)chloride.
B)pentamminechlorocobalt(II)chloride.
C)pentamminechlorocobalt(III)chloride.
D)pentamminechlorocobaltate(III)chloride.
E)pentamminedichlorocobalt(III).
A)chloropentamminecobalt(III)chloride.
B)pentamminechlorocobalt(II)chloride.
C)pentamminechlorocobalt(III)chloride.
D)pentamminechlorocobaltate(III)chloride.
E)pentamminedichlorocobalt(III).

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
37
What is the coordination number for Ni(CN)42-?
A)2
B)3
C)6
D)4
E)5
A)2
B)3
C)6
D)4
E)5

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
38
The correct name for [Co(NH3)6][Cr(CN)6] is __________
A)cobaltammine chromiumcyanide.
B)hexamminecobalt(III)hexacyanochromate(II).
C)hexamminecobaltate(III)hexacyanochromium(III).
D)hexamminecobalt(III)hexacyanochromate(III).
E)hexamminecobalt(II)hexacyanochromate(III).
A)cobaltammine chromiumcyanide.
B)hexamminecobalt(III)hexacyanochromate(II).
C)hexamminecobaltate(III)hexacyanochromium(III).
D)hexamminecobalt(III)hexacyanochromate(III).
E)hexamminecobalt(II)hexacyanochromate(III).

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
39
The correct formula for the complex ion tetracyanoplatinate(II)is __________
A)[Pt(CN)42+].
B)[Pt(CN)4]2-.
C)Pt(CN)4.
D)[Pt2(CN)4].
E)4[Pt(CN)]+.
A)[Pt(CN)42+].
B)[Pt(CN)4]2-.
C)Pt(CN)4.
D)[Pt2(CN)4].
E)4[Pt(CN)]+.

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
40
What is the correct formula for hexamminecobalt(III)nitrite?
A)[Co(NH3)6](NO3)3
B)Co(NH3)6(NO2)3
C)[Co(NH3)6](NO2)3
D)[Co(NH3)6](NO2)2
E)[Co(NH3)6](NO3)2
A)[Co(NH3)6](NO3)3
B)Co(NH3)6(NO2)3
C)[Co(NH3)6](NO2)3
D)[Co(NH3)6](NO2)2
E)[Co(NH3)6](NO3)2

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
41
Oxalic acid is a dicarboxylic acid with the structure HOOCCOOH.How many coordination sites does the ion (OOCCOO)2- have?
A)2
B)3
C)4
D)1
E)5
A)2
B)3
C)4
D)1
E)5

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
42
Which of the following is a polydentate ligand?
A)NO2-
B)OH-
C)H2O
D)C2O42-
E)CN-
A)NO2-
B)OH-
C)H2O
D)C2O42-
E)CN-

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
43
Which one of the following compounds is called diamminetriaquahydroxochromium(II)nitrate?
A)[Cr(NH3)2(H2O)3(OH)](NO3)2
B)[Cr(NH3)2(H2O)3(OH)]NO3
C)[Cr(NH3)2(H2O)3](OH)NO3
D)[Cr(NH3)2(OH)]NO3 • 3H2O
E)[Cr(NH3)2NO3]OH(aq)
A)[Cr(NH3)2(H2O)3(OH)](NO3)2
B)[Cr(NH3)2(H2O)3(OH)]NO3
C)[Cr(NH3)2(H2O)3](OH)NO3
D)[Cr(NH3)2(OH)]NO3 • 3H2O
E)[Cr(NH3)2NO3]OH(aq)

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
44
When a molecule of ethylenediamine replaces two molecules of NH3 in Co(NH3)63+,the entropy of the system __________
A)increases.
B)decreases.
C)remains the same.
D)cannot be determined.
E)is irrelevant.
A)increases.
B)decreases.
C)remains the same.
D)cannot be determined.
E)is irrelevant.

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
45
Which atoms on EDTA bond to metal ions?
A)carbon and oxygen
B)carbon and nitrogen
C)oxygen and nitrogen
D)nitrogen and hydrogen
E)oxygen and hydrogen
A)carbon and oxygen
B)carbon and nitrogen
C)oxygen and nitrogen
D)nitrogen and hydrogen
E)oxygen and hydrogen

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
46
The reaction,Cr(NH3)63+(aq)+ 3 en(aq) Cr(en)33+(aq)+ 6 NH3(aq),where en represents ethylenediamine,has a small value for the enthalpy change, Hrxn,yet the free-energy change is large because __________
A)the reaction rate is fast.
B)the entropy change is large and positive.
C)the enthalpy change is large enough to matter.
D)the entropy change is large and negative.
E)ethylene diamine has amino groups that are stronger bases than ammonia.
A)the reaction rate is fast.
B)the entropy change is large and positive.
C)the enthalpy change is large enough to matter.
D)the entropy change is large and negative.
E)ethylene diamine has amino groups that are stronger bases than ammonia.

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
47
Which is the correct formula for pentaamminecyanocobaltate(III)chloride?
A)[Co(NH3)5CN]Cl2
B)[Co(NH3)5CN]Cl3
C)[Co(NH3)5Cl](CN)2
D)[Co(NH3)5CNO]Cl2
E)[Co(NH3)5CNO]Cl3
A)[Co(NH3)5CN]Cl2
B)[Co(NH3)5CN]Cl3
C)[Co(NH3)5Cl](CN)2
D)[Co(NH3)5CNO]Cl2
E)[Co(NH3)5CNO]Cl3

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
48
The greater affinity of metal ions for polydentate ligands than for monodentate ligands is known as the __________ effect.
A)dentate
B)ligand
C)chelate
D)Lewis base
E)Lewis acid
A)dentate
B)ligand
C)chelate
D)Lewis base
E)Lewis acid

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
49
Nickel ions,Ni2+,should have the greatest affinity for __________
A)water.
B)ammonia.
C)chloride.
D)diethylenetriamine.
E)oxalate.
A)water.
B)ammonia.
C)chloride.
D)diethylenetriamine.
E)oxalate.

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
50
The structure of the acetylacetonate ligand is shown.How many coordination sites does it have? 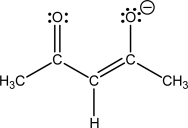
A)5
B)4
C)3
D)2
E)1

A)5
B)4
C)3
D)2
E)1

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
51
The correct formula for ammonium tetracyanoplatinate(II)is __________
A)(NH3)2[Pt(CN)4].
B)(NH4)2[Pt(CN)4].
C)(NH4)2Pt(CN)4.
D)(NH4)2[Pt(CN)6].
E)NH4[Pt(CN)4].
A)(NH3)2[Pt(CN)4].
B)(NH4)2[Pt(CN)4].
C)(NH4)2Pt(CN)4.
D)(NH4)2[Pt(CN)6].
E)NH4[Pt(CN)4].

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
52
Ethylenediamine is an example of a __________ ligand.
A)tridentate
B)hexadentate
C)bidentate
D)pentadentate
E)hydrocarbon
A)tridentate
B)hexadentate
C)bidentate
D)pentadentate
E)hydrocarbon

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
53
Which is the correct formula for potassium hexacyanoferrate(II)?
A)K[Fe(CN)2]
B)K2[Fe(CN)2]
C)K3[Fe(CN)6]
D)K4[Fe(CN)6]
E)K6[Fe(CN)6]
A)K[Fe(CN)2]
B)K2[Fe(CN)2]
C)K3[Fe(CN)6]
D)K4[Fe(CN)6]
E)K6[Fe(CN)6]

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
54
The reaction,Cr(NH3)63+(aq)+ 3 en(aq) Cr(en)33+(aq)+ 6 NH3(aq),where en represents ethylenediamine,has a small value for the enthalpy change, Hrxn,yet the equilibrium constant for this reaction is large because __________
A)the reaction rate is fast.
B)the entropy change is large and positive.
C)the enthalpy change is large enough to matter.
D)the entropy change is large and negative.
E)the equilibrium constant does not depend on the enthalpy change.
A)the reaction rate is fast.
B)the entropy change is large and positive.
C)the enthalpy change is large enough to matter.
D)the entropy change is large and negative.
E)the equilibrium constant does not depend on the enthalpy change.

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
55
Given the following data,calculate the approximate value of the equilibrium constant at 298 K for the reaction,Ni(NH3)62+(aq)+ 3 en(aq) Ni(en)32+(aq)+ 6 NH3(aq),where en represents ethylenediamine.
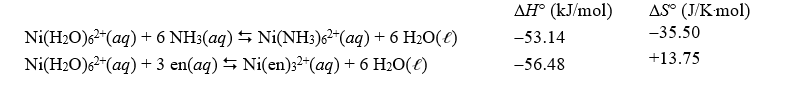
A)1.90 10-2
B)52.7
C)3.77
D)1.44 103
E)6.94 10-4
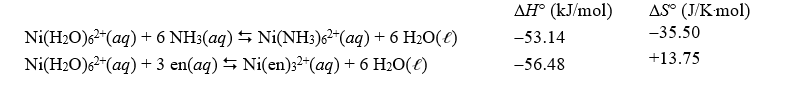
A)1.90 10-2
B)52.7
C)3.77
D)1.44 103
E)6.94 10-4

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
56
How many chelation sites and donor groups does EDTA have?
A)2
B)10
C)6
D)4
E)5
A)2
B)10
C)6
D)4
E)5

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
57
Which of the following is a chelation agent?
A)EDTA
B)Cl-
C)NH2
D)SCN-
E)CN-
A)EDTA
B)Cl-
C)NH2
D)SCN-
E)CN-

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
58
The interaction of a metal ion with a ligand having multiple atoms able to form coordinate covalent bonds is called __________
A)coordinating.
B)clawing.
C)precipitation.
D)chelation.
E)neutralization.
A)coordinating.
B)clawing.
C)precipitation.
D)chelation.
E)neutralization.

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
59
EDTA is an example of a __________ ligand.
A)tridentate
B)hexadentate
C)tetradentate
D)pentadentate
E)hydrocarbon
A)tridentate
B)hexadentate
C)tetradentate
D)pentadentate
E)hydrocarbon

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
60
What is the correct formula for sodium tetracyanodihydroxovanadate(II)?
A)Na2[V(CN)4(OH)2]
B)Na3[V(CN)4(OH)2]
C)Na4[V(CN)4(OH)2]
D)Na[V(CN)4(OH)2]
E)Na3[V(CN)4(OH)]
A)Na2[V(CN)4(OH)2]
B)Na3[V(CN)4(OH)2]
C)Na4[V(CN)4(OH)2]
D)Na[V(CN)4(OH)2]
E)Na3[V(CN)4(OH)]

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
61
Co(NH3)63+(aq)absorbs light in the blue region of the spectrum.The color of a solution containing this ion is __________
A)blue.
B)blue-violet.
C)violet.
D)yellow-orange.
E)red.
A)blue.
B)blue-violet.
C)violet.
D)yellow-orange.
E)red.

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
62
When the five 3d orbitals on a metal ion experience an octahedral field in a complex,they split into __________ energy levels.
A)two
B)three
C)four
D)five
E)six
A)two
B)three
C)four
D)five
E)six

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
63
Which of the following complexes has the smallest crystal field splitting of the d orbitals?
A)CoF63-
B)Co(CN)63-
C)CoBr63-
D)Co(H2O)63+
E)CoI63-
A)CoF63-
B)Co(CN)63-
C)CoBr63-
D)Co(H2O)63+
E)CoI63-

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
64
Transition metals with __________ tend to form square planar complexes.
A)d 8 and d 9 configurations
B)d 1 and d 2 configurations
C)half-filled d orbitals
D)high spin
E)no d electrons
A)d 8 and d 9 configurations
B)d 1 and d 2 configurations
C)half-filled d orbitals
D)high spin
E)no d electrons

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
65
Complex ions with different ligands have different colors because the ligands __________
A)are different colors.
B)affect the energy levels of the lone-pair electrons on the metal.
C)have different energies for their bonding electrons.
D)affect the energy levels of the metal d orbitals.
E)have different energies for their lone-pair electrons.
A)are different colors.
B)affect the energy levels of the lone-pair electrons on the metal.
C)have different energies for their bonding electrons.
D)affect the energy levels of the metal d orbitals.
E)have different energies for their lone-pair electrons.

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
66
Which of the following ligands will cause the smallest crystal field splitting of the d orbitals in an octahedral complex?
A)H2O
B)Br-
C)NH3
D)CN-
E)I-
A)H2O
B)Br-
C)NH3
D)CN-
E)I-

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
67
The dz2 and dx2-y2 orbitals are higher in energy than the dxy,dxz,and dyz orbitals in an octahedral complex because these two orbitals __________
A)do not point directly at ligands.
B)point directly at ligands.
C)occupy larger volumes than the other three orbitals.
D)are in the same plane and repel one another.
E)occupy smaller volumes than the other three orbitals.
A)do not point directly at ligands.
B)point directly at ligands.
C)occupy larger volumes than the other three orbitals.
D)are in the same plane and repel one another.
E)occupy smaller volumes than the other three orbitals.

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
68
Which of the following complexes has the largest crystal field splitting of the d orbitals?
A)CoF63-
B)Co(CN)63-
C)CoBr63-
D)Co(H2O)63+
E)CoI63-
A)CoF63-
B)Co(CN)63-
C)CoBr63-
D)Co(H2O)63+
E)CoI63-

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
69
In a tetrahedral crystal field,__________
A)dxy,dxz,and dyz are lower in energy than dx2-y2 and dz2.
B)dx2-y2 and dz2 are lower in energy than dxy,dxz,and dyz.
C)dxy,dxz,dyz,dx2-y2,and dz2 all have different energies.
D)dxy,dxz,dyz,dx2-y22,and dz2 all have the same energy.
E)dxy,dxz,and dyz have the same energy,and dx2-y2 and dz2 have different energies.
A)dxy,dxz,and dyz are lower in energy than dx2-y2 and dz2.
B)dx2-y2 and dz2 are lower in energy than dxy,dxz,and dyz.
C)dxy,dxz,dyz,dx2-y2,and dz2 all have different energies.
D)dxy,dxz,dyz,dx2-y22,and dz2 all have the same energy.
E)dxy,dxz,and dyz have the same energy,and dx2-y2 and dz2 have different energies.

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
70
Which statement describing the splitting of metal d orbitals in an octahedral field is NOT correct?
A)The d orbitals are split into two energy levels.
B)One energy level contains three orbitals.
C)One energy level contains two orbitals.
D)The dxy,dxz,and dyz orbitals have the lowest energy.
E)Photons cannot produce electronic transitions from one energy level to another.
A)The d orbitals are split into two energy levels.
B)One energy level contains three orbitals.
C)One energy level contains two orbitals.
D)The dxy,dxz,and dyz orbitals have the lowest energy.
E)Photons cannot produce electronic transitions from one energy level to another.

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
71
Which d orbitals have a higher energy in an octahedral complex?
A)dxy and dxz
B)dz2 and dyz
C)dz2 and dx2-y2
D)dx2-y2 and dxz
E)dxy,dxz,and dyz
A)dxy and dxz
B)dz2 and dyz
C)dz2 and dx2-y2
D)dx2-y2 and dxz
E)dxy,dxz,and dyz

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
72
Which 3d orbitals have a higher energy in an octahedral crystal field?
A)dxy and dxz
B)dz2 and dyz
C)dz2 and dx2-y2
D)dx2-y2 and dxz
E)dxy,dxz,and dyz
A)dxy and dxz
B)dz2 and dyz
C)dz2 and dx2-y2
D)dx2-y2 and dxz
E)dxy,dxz,and dyz

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
73
A solution containing the ion CoF63- has an absorption maximum at max = 680 nm.The color of this solution is __________
A)red.
B)green.
C)blue.
D)orange.
E)violet.
A)red.
B)green.
C)blue.
D)orange.
E)violet.

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
74
As the value of the crystal field splitting, o,increases from one complex ion to another,__________
A)the wavelength of absorbed light increases.
B)the wavelength of absorbed light decreases.
C)the number of d electrons increases.
D)the number of d electrons decreases.
E)the color of the solution shifts from red toward blue.
A)the wavelength of absorbed light increases.
B)the wavelength of absorbed light decreases.
C)the number of d electrons increases.
D)the number of d electrons decreases.
E)the color of the solution shifts from red toward blue.

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
75
The dxy,dxz,and dyz orbitals are lower in energy than the dz2 and dx2-y2 orbitals in an octahedral complex because these three orbitals __________
A)do not point directly at ligands.
B)point directly at ligands.
C)occupy larger volumes than the other two orbitals.
D)are in the same plane and repel one another.
E)occupy smaller volumes than the other two orbitals.
A)do not point directly at ligands.
B)point directly at ligands.
C)occupy larger volumes than the other two orbitals.
D)are in the same plane and repel one another.
E)occupy smaller volumes than the other two orbitals.

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
76
Transition metal ions often absorb visible light in promoting electrons from a ground state to an excited state.Why doesn't aluminum(III)ion have this property?
A)It does,and this fact accounts for the color of some gemstones.
B)Aluminum(III)has a noble gas configuration-that of argon.
C)Aluminum(III)has a noble gas configuration-that of neon.
D)Aluminum(III)has a filled d subshell.
E)The crystal field splitting of the d orbitals in aluminum(III)is very large.
A)It does,and this fact accounts for the color of some gemstones.
B)Aluminum(III)has a noble gas configuration-that of argon.
C)Aluminum(III)has a noble gas configuration-that of neon.
D)Aluminum(III)has a filled d subshell.
E)The crystal field splitting of the d orbitals in aluminum(III)is very large.

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
77
Given that the observed absorption maximum for the complex ion CoF63- in solution is max = 700 nm,what is the value of the crystal field splitting, o?
A)3 10-19 J
B)3 10-28 J
C)7 1030 J
D)3 10-36 J
E)7 10-19 J
A)3 10-19 J
B)3 10-28 J
C)7 1030 J
D)3 10-36 J
E)7 10-19 J

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
78
Which d orbital(s)is/are highest in energy in a tetrahedral complex?
A)dz2
B)dxy,dxz,and dyz
C)dz2 and dx2-y2
D)dx2-y2
E)dxy and dxz
A)dz2
B)dxy,dxz,and dyz
C)dz2 and dx2-y2
D)dx2-y2
E)dxy and dxz

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
79
Which of the following ligands will cause the largest crystal field splitting of the d orbitals in an octahedral complex?
A)H2O
B)Br-
C)NH3
D)CN-
E)I-
A)H2O
B)Br-
C)NH3
D)CN-
E)I-

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck
80
Crystal field theory describes __________
A)how ligands cause the metal d orbitals to have different energies.
B)the covalent bonding in transition metal complexes.
C)how d orbitals change their shapes when ligands are present.
D)how metal s,p,and d orbitals are hybridized to bond with ligands.
E)the molecular orbitals that describe the bonding in transition metal complexes.
A)how ligands cause the metal d orbitals to have different energies.
B)the covalent bonding in transition metal complexes.
C)how d orbitals change their shapes when ligands are present.
D)how metal s,p,and d orbitals are hybridized to bond with ligands.
E)the molecular orbitals that describe the bonding in transition metal complexes.

Unlock Deck
Unlock for access to all 153 flashcards in this deck.
Unlock Deck
k this deck



