Deck 9: Alkynes
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question

Unlock Deck
Sign up to unlock the cards in this deck!
Unlock Deck
Unlock Deck
1/15
Play
Full screen (f)
Deck 9: Alkynes
1
What is the IUPAC name of the following compound?

A) 5-propyl-3-heptyne
B) 5-isopropyl-3-heptyne
C) 5-ethyl-3-octyne
D) 4-ethyl-5-octyne

A) 5-propyl-3-heptyne
B) 5-isopropyl-3-heptyne
C) 5-ethyl-3-octyne
D) 4-ethyl-5-octyne
5-ethyl-3-octyne
2
Which of the following describes the orbital overlap of the sigma bond of 1-butyne, shown below?

A)
B)
C)
D)

A)
B)
C)
D)
3
Select the best base to quantitatively remove a proton from acetylene.
A)
B)
C)
D)
A)
B)
C)
D)
4
Predict the major product(s) in the reactions below.
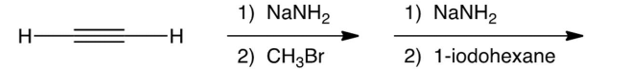
A) 1-nonyne
B) 2-nonyne
C) cis-2-nonene
D) trans-2-nonene

A) 1-nonyne
B) 2-nonyne
C) cis-2-nonene
D) trans-2-nonene

Unlock Deck
Unlock for access to all 15 flashcards in this deck.
Unlock Deck
k this deck
5
Which one of the following alkynes gives a single ketone in the acid-catalyzed hydration of each?
A) 2-decyne
B) 3-decyne
C) 4-decyne
D) 5-decyne
A) 2-decyne
B) 3-decyne
C) 4-decyne
D) 5-decyne

Unlock Deck
Unlock for access to all 15 flashcards in this deck.
Unlock Deck
k this deck
6
Which sequence of reactions works best in synthesizing cis-3-nonene?

A) A
B) B
C) C
D) D

A) A
B) B
C) C
D) D

Unlock Deck
Unlock for access to all 15 flashcards in this deck.
Unlock Deck
k this deck
7
Which sequence of reactions below works best in carrying out the following conversion?
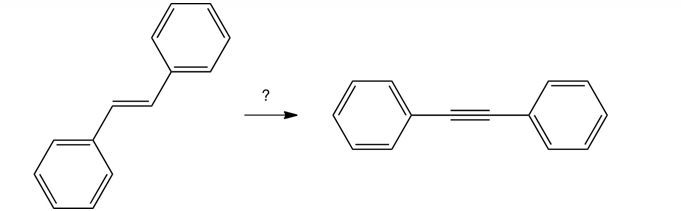
A) (1) excess
B) (1) (2) excess
C) (1) (2) excess
D) (1) (cat.) (2) excess

A) (1) excess
B) (1) (2) excess
C) (1) (2) excess
D) (1) (cat.) (2) excess

Unlock Deck
Unlock for access to all 15 flashcards in this deck.
Unlock Deck
k this deck
8
Which of the following is the enol intermediate in the acid-catalyzed addition of water to propyne?
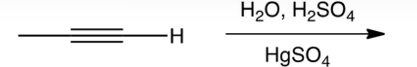
A)
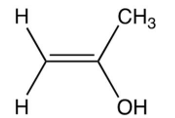
B)
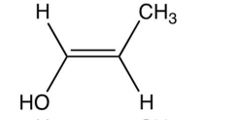
C)
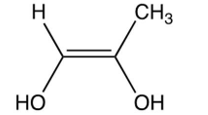
D)
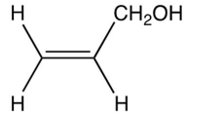
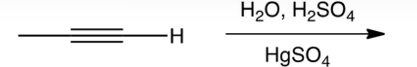
A)

B)

C)

D)


Unlock Deck
Unlock for access to all 15 flashcards in this deck.
Unlock Deck
k this deck
9
Identify compound .
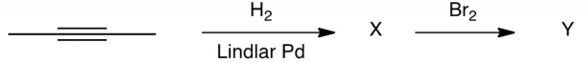
A) 2-bromobutane
B) meso-2,3-dibromobutane
C) racemic (2R,3R) and (2S,3S)-2,3-dibromobutane
D) 2,3-dibromo-2-butene

A) 2-bromobutane
B) meso-2,3-dibromobutane
C) racemic (2R,3R) and (2S,3S)-2,3-dibromobutane
D) 2,3-dibromo-2-butene

Unlock Deck
Unlock for access to all 15 flashcards in this deck.
Unlock Deck
k this deck
10
What is the major product of the reaction shown below?
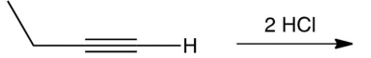
A) 1,1-dichlorobutane
B) 2,2-dichlorobutane
C) 1,2-dichlorobutane
D) 1,12,2-tetrachlorobutane
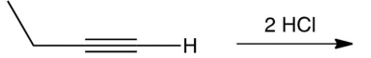
A) 1,1-dichlorobutane
B) 2,2-dichlorobutane
C) 1,2-dichlorobutane
D) 1,12,2-tetrachlorobutane

Unlock Deck
Unlock for access to all 15 flashcards in this deck.
Unlock Deck
k this deck
11
Why can't methanol, , be used as a solvent for sodium amide, ?
A) Sodium amide is nonpolar and methanol is polar.
B) Sodium amide is polar and methanol is nonpolar.
C) Sodium amide does an acid-base reaction with methanol.
D) There would be no ion-dipole attractive forces between the two compounds.
A) Sodium amide is nonpolar and methanol is polar.
B) Sodium amide is polar and methanol is nonpolar.
C) Sodium amide does an acid-base reaction with methanol.
D) There would be no ion-dipole attractive forces between the two compounds.

Unlock Deck
Unlock for access to all 15 flashcards in this deck.
Unlock Deck
k this deck
12
Ozonolysis of an alkyne gave the two compounds shown below. What is the IUPAC name of the original alkyne?
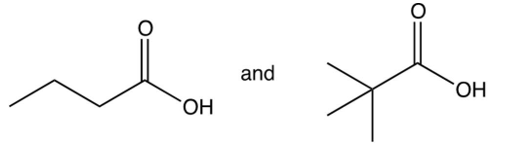
and
A) 2,2-dimethyl-3-octyne
B) 2,2-dimethyl-3-heptyne
C) 3,3-dimethyl-4-octyne
D) 6,6-dimethyl-3-heptyne

and
A) 2,2-dimethyl-3-octyne
B) 2,2-dimethyl-3-heptyne
C) 3,3-dimethyl-4-octyne
D) 6,6-dimethyl-3-heptyne

Unlock Deck
Unlock for access to all 15 flashcards in this deck.
Unlock Deck
k this deck
13
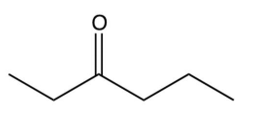
Which of the following ketones cannot be made by the acid-catalyzed hydration of an alkyne?
A)
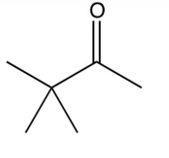
B)
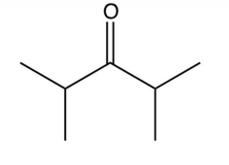
C)
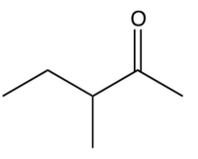
D)
A)

B)

C)

D)


Unlock Deck
Unlock for access to all 15 flashcards in this deck.
Unlock Deck
k this deck
14
Is the proposed synthesis of cyclohexanone below likely to work? If not, why not?
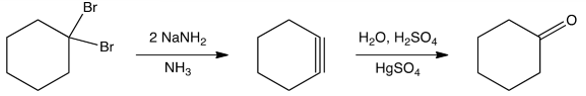
A) Yes, it would work.
B) No, you would need to start with 1, 2-dibromocyclohexane.
C) No, cyclohexyne will not form.
D) No, the enol of cyclohexanone cannot be formed from cyclohexyne.

A) Yes, it would work.
B) No, you would need to start with 1, 2-dibromocyclohexane.
C) No, cyclohexyne will not form.
D) No, the enol of cyclohexanone cannot be formed from cyclohexyne.

Unlock Deck
Unlock for access to all 15 flashcards in this deck.
Unlock Deck
k this deck
15
What major product results from this reaction?
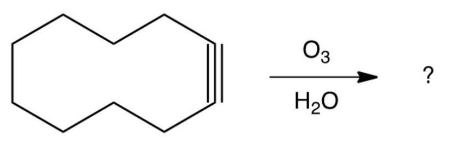
A)
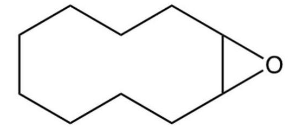
B)
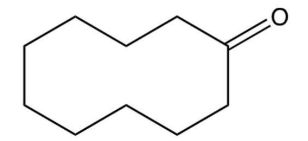
C)
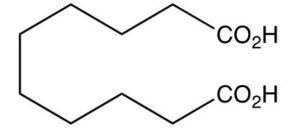
D)
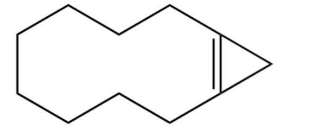

A)

B)

C)

D)


Unlock Deck
Unlock for access to all 15 flashcards in this deck.
Unlock Deck
k this deck



