Deck 16: Ethers, Epoxides, and Sulfides
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question

Unlock Deck
Sign up to unlock the cards in this deck!
Unlock Deck
Unlock Deck
1/13
Play
Full screen (f)
Deck 16: Ethers, Epoxides, and Sulfides
1
Which of the following compounds is the most soluble in water?
A)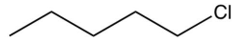
B)
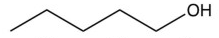
C)
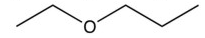
D)
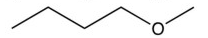
A)

B)

C)

D)


2
Which of the following is the correct name of the compound below?
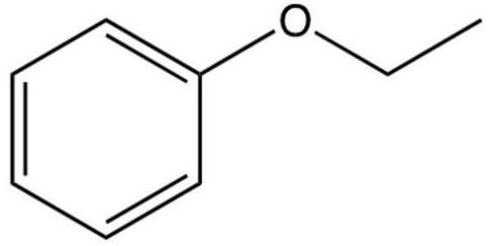
A) benzyl ethyl ether
B) ethylbenzene ether
C) ethyl phenyl ether
D) ethoxy phenoxy ether

A) benzyl ethyl ether
B) ethylbenzene ether
C) ethyl phenyl ether
D) ethoxy phenoxy ether
ethyl phenyl ether
3
Match the boiling points with the following three isomers of .
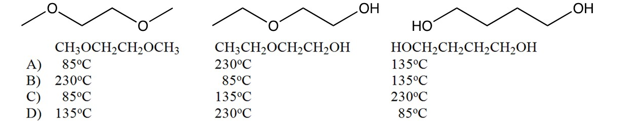
A) A
B) B
C) C
D) D

A) A
B) B
C) C
D) D
C
4
What is the product of the following reaction?
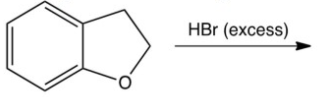
A)
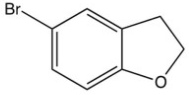
B)
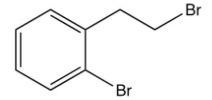
C)
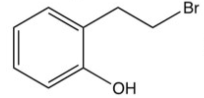
D)

A)

B)

C)

D)


Unlock Deck
Unlock for access to all 13 flashcards in this deck.
Unlock Deck
k this deck
5
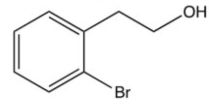
Which of the following is not an intermediate in the reaction below?
A)
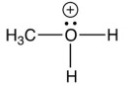
B)
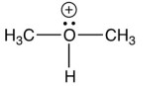
C)
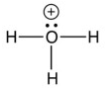
D) they are all intermediates.
A)

B)

C)

D) they are all intermediates.

Unlock Deck
Unlock for access to all 13 flashcards in this deck.
Unlock Deck
k this deck
6
Which one of the following reactions makes the cyclic ether shown below?
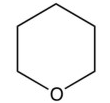
A)
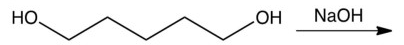
B)
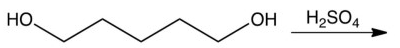
C)
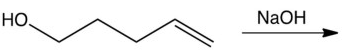
D)
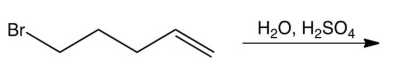

A)

B)

C)

D)


Unlock Deck
Unlock for access to all 13 flashcards in this deck.
Unlock Deck
k this deck
7
What is the product of the following reaction? 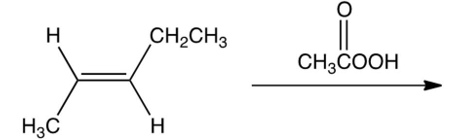
A)
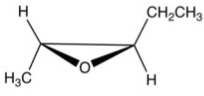
B)
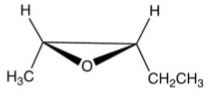
C)
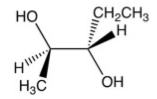
D)
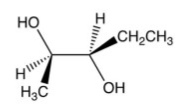

A)

B)

C)

D)


Unlock Deck
Unlock for access to all 13 flashcards in this deck.
Unlock Deck
k this deck
8
The reaction shown below can be described as an:
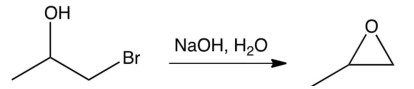
A) acid-base reaction followed by an intramolecular Williamson ether synthesis.
B) acid-base reaction followed by an intramolecular reaction.
C) E2 reaction followed by an addition reaction to a double bond.
D) reaction followed by an intramolecular Williamson ether synthesis.
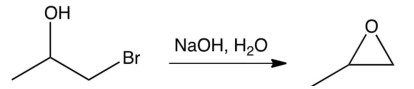
A) acid-base reaction followed by an intramolecular Williamson ether synthesis.
B) acid-base reaction followed by an intramolecular reaction.
C) E2 reaction followed by an addition reaction to a double bond.
D) reaction followed by an intramolecular Williamson ether synthesis.

Unlock Deck
Unlock for access to all 13 flashcards in this deck.
Unlock Deck
k this deck
9
What is the final product of the following sequence of reactions?
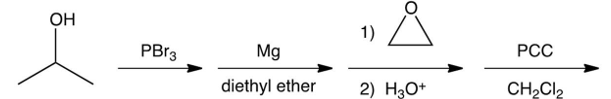
A)
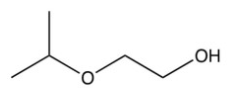
B)
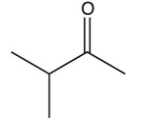
C)
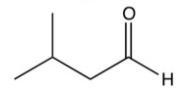
D)
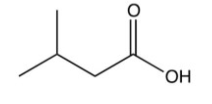

A)

B)

C)

D)


Unlock Deck
Unlock for access to all 13 flashcards in this deck.
Unlock Deck
k this deck
10
What is the product of the reactions below?
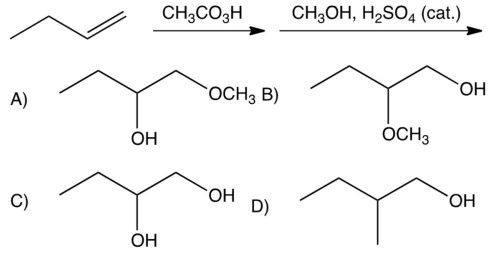
A) A
B) B
C) C
D) D

A) A
B) B
C) C
D) D

Unlock Deck
Unlock for access to all 13 flashcards in this deck.
Unlock Deck
k this deck
11
Which of the following epoxides is formed when is added to the optically active halohydrin shown below? 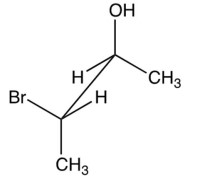
A) trans-(2S,3S)-2,3-dimethyloxirane
B) trans-(2R,3R)-2,3-dimethyloxirane
C) 2,2-dimethyloxirane
D) meso-2,3-dimethyloxirane

A) trans-(2S,3S)-2,3-dimethyloxirane
B) trans-(2R,3R)-2,3-dimethyloxirane
C) 2,2-dimethyloxirane
D) meso-2,3-dimethyloxirane

Unlock Deck
Unlock for access to all 13 flashcards in this deck.
Unlock Deck
k this deck
12
Ethylene glycol reacts in acid to form a cyclic compound with the formula of . Which one of the following is this cyclic compound?
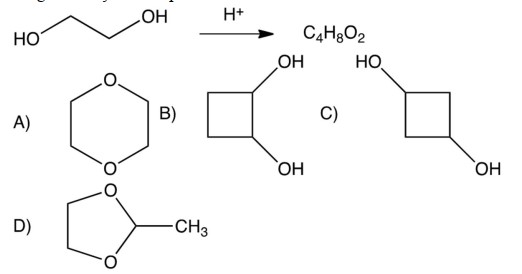
A) A
B) B
C) C
D) D

A) A
B) B
C) C
D) D

Unlock Deck
Unlock for access to all 13 flashcards in this deck.
Unlock Deck
k this deck
13
What results when dimethylsulfide is treated with sodium periodate?
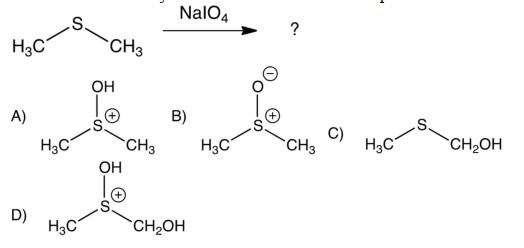
A) A
B) B
C) C
D) D

A) A
B) B
C) C
D) D

Unlock Deck
Unlock for access to all 13 flashcards in this deck.
Unlock Deck
k this deck



