Deck 9: The World of Polymers and Plastics
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question

Unlock Deck
Sign up to unlock the cards in this deck!
Unlock Deck
Unlock Deck
1/84
Play
Full screen (f)
Deck 9: The World of Polymers and Plastics
1
What do Lycra ,Gore-Tex ,Saran™,Teflon ,and Formica have in common?
A)They are all brands produced by Dupont.
B)They are all patents owned by Charles Watson.
C)They are all specialized synthetic polymers.
D)They are all approved for use by the FDA.
A)They are all brands produced by Dupont.
B)They are all patents owned by Charles Watson.
C)They are all specialized synthetic polymers.
D)They are all approved for use by the FDA.
They are all specialized synthetic polymers.
2
The "Big Six" polymers are all classified as thermoplastics because they
A)are all used as insulators.
B)can be melted and shaped.
C)are all crystalline.
D)are all amorphous.
A)are all used as insulators.
B)can be melted and shaped.
C)are all crystalline.
D)are all amorphous.
can be melted and shaped.
3
Which of these synthetic polymers is most closely related to the natural protein polymers?
A)polyester
B)nylon
C)polystyrene
D)polyvinyl chloride
A)polyester
B)nylon
C)polystyrene
D)polyvinyl chloride
nylon
4
Which of these is vinyl chloride?
A)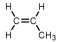
B)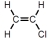
C)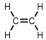
D)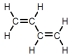
A)

B)

C)

D)


Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
5
Polyvinyl chloride (PVC)is used in
A)water bottles.
B)CD cases.
C)milk cartons.
D)plumbing pipe.
A)water bottles.
B)CD cases.
C)milk cartons.
D)plumbing pipe.

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
6
Which polymer is incorrectly matched with its use?
A)LDPE-plastic bag
B)HDPE-milk jug
C)PS-contact lenses
D)PET-soda bottles
A)LDPE-plastic bag
B)HDPE-milk jug
C)PS-contact lenses
D)PET-soda bottles

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
7
This is a schematic representation of the two different forms of polyethylene. 
Which of the following statements is true?
A)I is HDPE and it has a lower melting point than II which is LDPE.
B)I is HDPE and it has a higher melting point than II which is LDPE.
C)I is LDPE and it has a lower melting point than II which is HDPE.
D)I is LDPE and it has a higher melting point than II which is HDPE.

Which of the following statements is true?
A)I is HDPE and it has a lower melting point than II which is LDPE.
B)I is HDPE and it has a higher melting point than II which is LDPE.
C)I is LDPE and it has a lower melting point than II which is HDPE.
D)I is LDPE and it has a higher melting point than II which is HDPE.

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
8
Polystyrene is used to make Styrofoam cups.For this usage,which property is most important?
A)good insulator
B)light weight
C)white and opaque
D)brittle
A)good insulator
B)light weight
C)white and opaque
D)brittle

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
9
Which would be classified as a "natural" polymer?
A)cellulose
B)polystyrene
C)polyester
D)nylon
A)cellulose
B)polystyrene
C)polyester
D)nylon

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
10
Which statement is false?
A)Microfiber is a polymer made into very fine threads.
B)Microfiber is used in clothing as a water repellent.
C)Microfiber is a good insulator.
D)Microfiber is a new type of polymer.
A)Microfiber is a polymer made into very fine threads.
B)Microfiber is used in clothing as a water repellent.
C)Microfiber is a good insulator.
D)Microfiber is a new type of polymer.

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
11
The molecules that are added to polymers to make them softer and more pliable are known as
A)monomers.
B)plasticizers.
C)pigments.
D)rubber.
A)monomers.
B)plasticizers.
C)pigments.
D)rubber.

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
12
This is the structure of propylene. 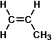
Which of these represents the "head-to-tail,head-to-tail" arrangement of monomers in polypropylene?
A)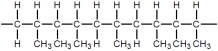
B)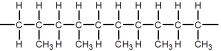
C)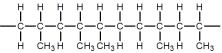
D)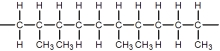

Which of these represents the "head-to-tail,head-to-tail" arrangement of monomers in polypropylene?
A)

B)

C)

D)


Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
13
Blowing agents are gases,or a compound that releases a gas,that are added to polymers in the manufacture of a foamed plastic.Which statement is not true?
A)At one time,CFC's were used as blowing agents.
B)Gaseous pentane is now often used.
C)The DOW 100% CO2 technology uses carbon monoxide.
D)The use of pentane is hazardous because it is highly flammable.
A)At one time,CFC's were used as blowing agents.
B)Gaseous pentane is now often used.
C)The DOW 100% CO2 technology uses carbon monoxide.
D)The use of pentane is hazardous because it is highly flammable.

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
14
Which would be classified as a "synthetic" polymer?
A)cellulose
B)wool
C)rubber
D)nylon
A)cellulose
B)wool
C)rubber
D)nylon

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
15
A monomer is
A)a single polymer chain.
B)a polymer made from only one component.
C)a small molecule used to make a polymer chain.
D)a polymer molecule that only contains a single element.
A)a single polymer chain.
B)a polymer made from only one component.
C)a small molecule used to make a polymer chain.
D)a polymer molecule that only contains a single element.

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
16
Nathaniel Wyeth developed plastic soft drink bottles.They are made from which polymer?
A)HDPE
B)PVC
C)PS
D)PET
A)HDPE
B)PVC
C)PS
D)PET

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
17
A polymer is a large molecule
A)made up of long chains of atoms held together by hydrogen bonds.
B)made up of long chains of atoms covalently bonded together.
C)made up of a large lattice structure held together by ionic bonds.
D)always made up of identical monomer units.
A)made up of long chains of atoms held together by hydrogen bonds.
B)made up of long chains of atoms covalently bonded together.
C)made up of a large lattice structure held together by ionic bonds.
D)always made up of identical monomer units.

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
18
Polystyrene is used to make
A)foam coffee cups and CD cases.
B)garbage bags and CDs.
C)shoes and garden hoses.
D)cell phones and contact lenses.
A)foam coffee cups and CD cases.
B)garbage bags and CDs.
C)shoes and garden hoses.
D)cell phones and contact lenses.

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
19
Some of the uses for polypropylene are yogurt containers,carpeting,luggage,and bottle caps.Which is not a property of polypropylene?
A)high melting point
B)very tough
C)transparent
D)resistant to oils
A)high melting point
B)very tough
C)transparent
D)resistant to oils

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
20
This is the structure of propylene. 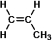
Which of these does not represent a possible arrangement of monomers within polypropylene?
A)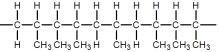
B)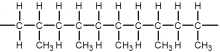
C)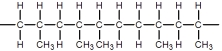
D)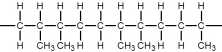

Which of these does not represent a possible arrangement of monomers within polypropylene?
A)

B)

C)

D)


Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
21
Which functional group do all monomers undergoing addition polymerization have in common?
A)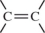
B)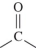
C)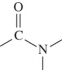
D)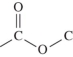
A)
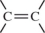
B)
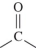
C)

D)


Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
22
In this molecule four different functional groups are circled. 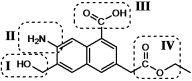
Which is the carboxylic acid functional group?
A)I
B)II
C)III
D)IV

Which is the carboxylic acid functional group?
A)I
B)II
C)III
D)IV

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
23
Which of these statements are correct for both addition and condensation polymerization?
I.Monomer units chemically bond together.
II.Product contains all the atoms of the starting material.
III.A second product,like water,is also formed during the polymerization process.
A)I only
B)I and II
C)II only
D)II and III
I.Monomer units chemically bond together.
II.Product contains all the atoms of the starting material.
III.A second product,like water,is also formed during the polymerization process.
A)I only
B)I and II
C)II only
D)II and III

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
24
Which bond represents the "peptide" bond? 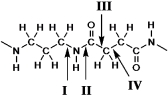
A)I
B)II
C)III
D)IV

A)I
B)II
C)III
D)IV

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
25
These are the two monomers used to synthesize Kevlar. 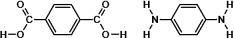
Which of the following represent the products formed after one condensation reaction between the monomers?
A)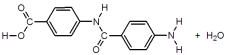
B)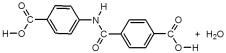
C)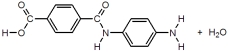
D)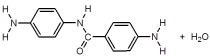

Which of the following represent the products formed after one condensation reaction between the monomers?
A)

B)

C)

D)


Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
26
Dacron is classified as a polyester.Which linkage is correct? (The gray areas represent the monomer backbones. )
A)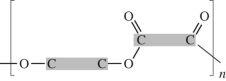
B)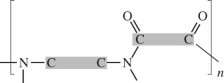
C)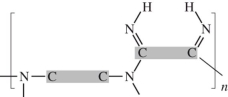
D)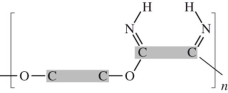
A)

B)

C)

D)


Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
27
The distinctive arrangements of atoms that impart characteristic chemical properties to the molecules that contain them are called
A)monomers.
B)functional groups.
C)radicals.
D)transition states.
A)monomers.
B)functional groups.
C)radicals.
D)transition states.

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
28
What do the letters HDPE represent?
A)high definition polyethylene
B)high density polyethylene
C)hard durable polyethylene
D)highly ductile polyethylene
A)high definition polyethylene
B)high density polyethylene
C)hard durable polyethylene
D)highly ductile polyethylene

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
29
Which of these statements are correct for addition polymerization only?
I.Monomer units chemically bond together.
II.Product contains all the atoms of the starting material.
III.There is generally just one monomer.
A)I only
B)I and II
C)II only
D)I and III
I.Monomer units chemically bond together.
II.Product contains all the atoms of the starting material.
III.There is generally just one monomer.
A)I only
B)I and II
C)II only
D)I and III

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
30
Which of these represents an amino acid?
A)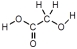
B)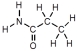
C)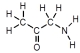
D)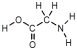
A)

B)

C)

D)


Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
31
Which equation correctly shows the first step in the formation of polyethylene terephthalate (PET)by the process of condensation polymerization?
A)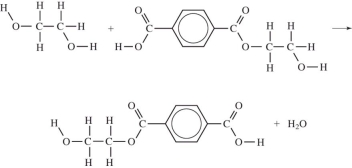
B)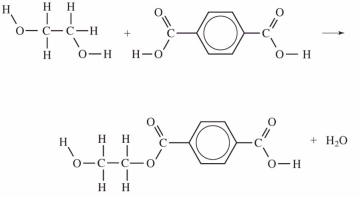
C)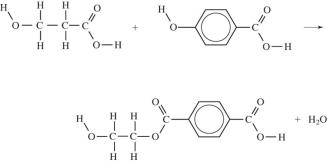
D)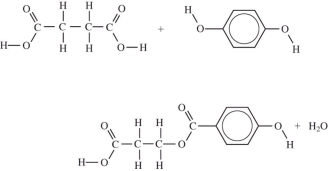
A)

B)

C)

D)


Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
32
Which equation below shows the addition polymerization of ethylene?
A)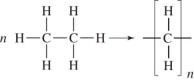
B)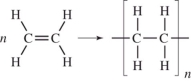
C)
D)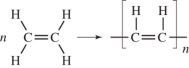
A)

B)

C)

D)


Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
33
A polymer that is made from two different monomers is known as a
A)dimer.
B)dipolymer.
C)copolymer.
D)multimer.
A)dimer.
B)dipolymer.
C)copolymer.
D)multimer.

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
34
How many carbon atoms are present in this monomer? 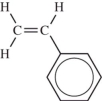
A)2
B)3
C)5
D)8

A)2
B)3
C)5
D)8

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
35
Which of these statements are correct for condensation polymerization only?
I.Monomer units chemically bond together.
II.Product contains all the atoms of the starting material.
III.A second product,like water,is also formed during the polymerization process.
A)I only
B)I and II
C)II only
D)I and III
I.Monomer units chemically bond together.
II.Product contains all the atoms of the starting material.
III.A second product,like water,is also formed during the polymerization process.
A)I only
B)I and II
C)II only
D)I and III

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
36
Consider the process of condensation polymerization.Which statement is false?
A)A small molecule like water is often formed as the monomers combine to form the polymer.
B)The product contains all of the atoms of the starting materials.
C)PET is formed using this process.
D)Nylon is formed using this process.
A)A small molecule like water is often formed as the monomers combine to form the polymer.
B)The product contains all of the atoms of the starting materials.
C)PET is formed using this process.
D)Nylon is formed using this process.

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
37
Which atoms does a hydroxyl functional group contain?
A)hydrogen and carbon
B)carbon and oxygen
C)hydrogen and oxygen
D)hydrogen and nitrogen
A)hydrogen and carbon
B)carbon and oxygen
C)hydrogen and oxygen
D)hydrogen and nitrogen

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
38
Which of these polymers is made by the process of condensation polymerization?
A)polyethylene
B)polystyrene
C)polyvinyl chloride
D)polyethylene terephthalate
A)polyethylene
B)polystyrene
C)polyvinyl chloride
D)polyethylene terephthalate

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
39
Which of these represents a portion of a polyester?
A)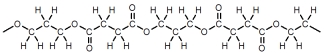
B)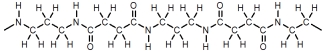
C)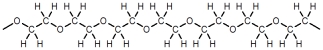
A)

B)

C)


Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
40
What do the "Big Six" polymers have in common?
A)They all are formed by addition polymerization.
B)They all are formed by condensation polymerization.
C)They all are thermoplastics (can be melted and shaped).
D)They all contain branched chains.
A)They all are formed by addition polymerization.
B)They all are formed by condensation polymerization.
C)They all are thermoplastics (can be melted and shaped).
D)They all contain branched chains.

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
41
Incineration of plastics can be used to generate energy.One of the "Big Six" plastics releases hydrochloric acid,HCl,when incinerated.Which one?
A)LDPE
B)PVC
C)PET
D)PS
A)LDPE
B)PVC
C)PET
D)PS

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
42
Polyvinyl chloride (PVC)is made from the monomer,CH2 
CHCl.Which would not be a possible arrangement in PVC?
A)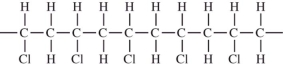
B)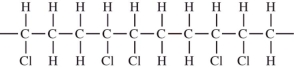
C)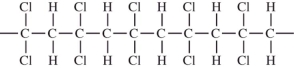
D)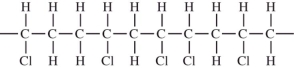

CHCl.Which would not be a possible arrangement in PVC?
A)

B)

C)

D)


Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
43
When nylon forms,adipic acid (a diacid)reacts with 1,6-diaminohexane to eliminate a water molecule and form the polymer.What type of polymerization is this and why?
A)addition,because the two monomers add together
B)condensation,because two monomers have condensed into one
C)addition,because all the atoms of the monomers are in the product
D)condensation,because water is eliminated
A)addition,because the two monomers add together
B)condensation,because two monomers have condensed into one
C)addition,because all the atoms of the monomers are in the product
D)condensation,because water is eliminated

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
44
When I and II react together to make nylon,what are the products of the first condensation reaction? 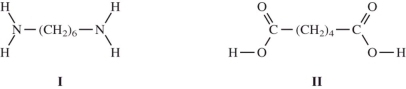
A)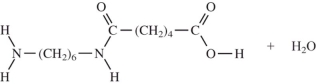
B)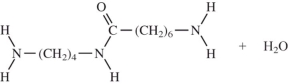
C)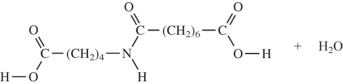
D)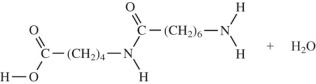
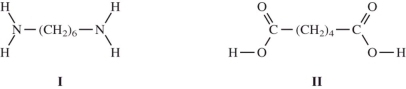
A)
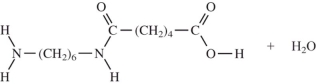
B)

C)

D)


Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
45
PET (polyethylene terephthalate)is classified as a polyester because
A)it contains many ester linkages.
B)its monomer has ester functional groups.
C)it is used to make bullet proof vests.
D)it is made by addition polymerization.
A)it contains many ester linkages.
B)its monomer has ester functional groups.
C)it is used to make bullet proof vests.
D)it is made by addition polymerization.

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
46
Incineration is a method used to dispose of used plastics.Which statement is false?
A)Plastics have higher energy content than coal.
B)Provided that it is carefully monitored and controlled,damage to the environment is limited.
C)Incineration of PVC may release hydrochloric acid,HCl,into the environment.
D)Incineration is the best method because it has no drawbacks.
A)Plastics have higher energy content than coal.
B)Provided that it is carefully monitored and controlled,damage to the environment is limited.
C)Incineration of PVC may release hydrochloric acid,HCl,into the environment.
D)Incineration is the best method because it has no drawbacks.

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
47
Which structure represents a peptide bond?
A)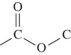
B)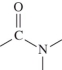
C)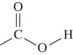
D)
A)

B)

C)

D)


Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
48
Proteins are polymers of amino acids held together by ______ bonds.
A)protein
B)peptide
C)ester
D)acid
A)protein
B)peptide
C)ester
D)acid

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
49
Which atoms are present in all proteins?
A)C,H,O,N
B)C,H,O,S
C)C,H,O only
D)C,H,N only
A)C,H,O,N
B)C,H,O,S
C)C,H,O only
D)C,H,N only

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
50
Why is crude oil currently the only raw material from which synthetic polymers can be made?
A)They cannot be made from anything else.
B)It is the most convenient and economical raw material.
C)Crude oil is a fossil fuel with an endless supply available.
D)Synthetic polymers are made to mimic natural ones.
A)They cannot be made from anything else.
B)It is the most convenient and economical raw material.
C)Crude oil is a fossil fuel with an endless supply available.
D)Synthetic polymers are made to mimic natural ones.

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
51
Adipic acid,one of the monomers used to make nylon,has a total of six carbon atoms with two carboxylic acid functional groups.Which structure is correct?
A)(CH2)6 COOH
B)HO (CH2)6 OH
C)HOOC (CH2)4 COOH
D)HOOC (CH2)6 NH2
A)(CH2)6 COOH
B)HO (CH2)6 OH
C)HOOC (CH2)4 COOH
D)HOOC (CH2)6 NH2

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
52
This is a representation of a plastic bag being stretched.Which statement best describes what is happening in the figure? 
A)Molecular chains shift from a random orientation to align parallel to each other in the direction of the pulling force.
B)During the pulling,the mixed-up polymer chains cross link to make the polymer stronger.
C)The pulling force creates branches,causing a switch from high density to low density polymer.
D)It is only a temporary distortion;once the pulling force stops,the plastic snaps back to the original orientation.

A)Molecular chains shift from a random orientation to align parallel to each other in the direction of the pulling force.
B)During the pulling,the mixed-up polymer chains cross link to make the polymer stronger.
C)The pulling force creates branches,causing a switch from high density to low density polymer.
D)It is only a temporary distortion;once the pulling force stops,the plastic snaps back to the original orientation.

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
53
One of the monomers used to make nylon is 1,6-diaminohexane (also known as hexamethylene diamine).How many amine ( NH2)groups does it have?
A)none
B)1
C)2
D)6
A)none
B)1
C)2
D)6

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
54
Will synthetic polymers always have to be made from crude oil?
A)Yes,because although other technologies exist,they are too costly for large scale use.
B)No,but there are no funds to research new technologies needed.
C)Yes,but more crude oil will have to be diverted from other uses.
D)No,new technologies are being developed to use bio-based raw materials.
A)Yes,because although other technologies exist,they are too costly for large scale use.
B)No,but there are no funds to research new technologies needed.
C)Yes,but more crude oil will have to be diverted from other uses.
D)No,new technologies are being developed to use bio-based raw materials.

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
55
Which raw material serves as the source for most polymers?
A)crude oil
B)natural gas
C)minerals
D)salts
A)crude oil
B)natural gas
C)minerals
D)salts

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
56
How are HDPE and LDPE different?
A)Only one is a member of the "Big Six."
B)They are made from different monomers.
C)One has branched chains while one is linear.
D)One is made by addition polymerization,one by condensation.
A)Only one is a member of the "Big Six."
B)They are made from different monomers.
C)One has branched chains while one is linear.
D)One is made by addition polymerization,one by condensation.

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
57
Nylon is classified as a polyamide because it contains which of the following linkages?
A)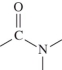
B)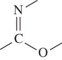
C)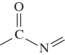
D)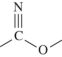
A)

B)

C)

D)


Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
58
The polymerization of ethylene is initiated by a free radical.Which structure represents a free radical formed during the polymerization?
A)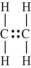
B)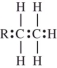
C)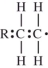
D)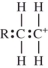
A)
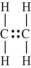
B)
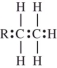
C)
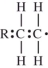
D)
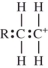

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
59
Every year about 100 billion pounds of plastic are produced in the United States (over 350 pounds for every woman,man,and child).What strategies may be employed to reduce the amount of plastics going into landfills?
A)incineration
B)biodegradation
C)recycling
D)All of these choices are correct.
A)incineration
B)biodegradation
C)recycling
D)All of these choices are correct.

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
60
Skin,hair,muscle,and enzymes are all made of protein,which is a natural polymer.Proteins are
A)polyesters.
B)polyethylenes.
C)polyamides.
D)polyalcohols.
A)polyesters.
B)polyethylenes.
C)polyamides.
D)polyalcohols.

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
61
This graph compares the recycling of plastics in Sweden and the United States. 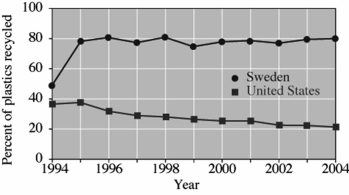
From this graph it is apparent that the percentage of plastics that Sweden recycled in 2004 was approximately ___ times the percentage that was recycled in the United States in that year.
A)80
B)8
C)6
D)4

From this graph it is apparent that the percentage of plastics that Sweden recycled in 2004 was approximately ___ times the percentage that was recycled in the United States in that year.
A)80
B)8
C)6
D)4

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
62
For recycling to be successful and self-sustaining which factors are important?
I.Plastics must be collected and sorted.
II.Recycled plastics must be made into products that consumers will buy.
III.Cities and towns have to impose fines for people who fail to recycle.
A)I only
B)II and III only
C)II only
D)I and II
I.Plastics must be collected and sorted.
II.Recycled plastics must be made into products that consumers will buy.
III.Cities and towns have to impose fines for people who fail to recycle.
A)I only
B)II and III only
C)II only
D)I and II

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
63
Superglue is made from this monomer,cyanoacrylate. 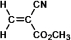
Which is a representation of the head-to-tail to head-to-tail arrangement of the polymer?
A)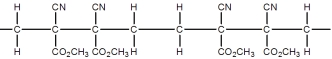
B)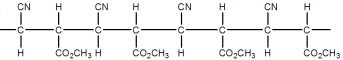
C)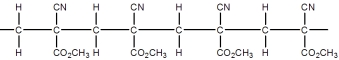
D)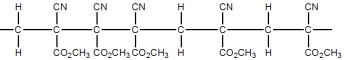

Which is a representation of the head-to-tail to head-to-tail arrangement of the polymer?
A)

B)

C)

D)


Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
64
Nathaniel Wyeth,developer of the plastic soft drink bottles,dreamed of the day when we will be able to melt down plastic bottles,reinforce the fibers and make car bodies.Then when we are done with the cars,melt down the car bodies and make bottles out of them.This would be an ultimate example of
A)biodegradation.
B)incineration.
C)recycling.
D)reuse.
A)biodegradation.
B)incineration.
C)recycling.
D)reuse.

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
65
The general formula for coal is C135H96NOS,the molecular formula for methane is CH4 and the molecular formula for dodecane is C12H26.Which will the heat of combustion of polyethylene be more similar to?
A)coal
B)methane
C)dodecane
D)hydrogen gas
A)coal
B)methane
C)dodecane
D)hydrogen gas

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
66
Which synthetic polymer is used to make shatter-resistant bottles and fleece fabric?
A)PET
B)HDPE
C)PVC
D)PS
A)PET
B)HDPE
C)PVC
D)PS

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
67
This represents a step in the addition polymerization of ethylene. 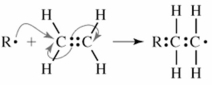
What do chemists call the species "R"?
A)a radical
B)a cation
C)an anion
D)an electron

What do chemists call the species "R"?
A)a radical
B)a cation
C)an anion
D)an electron

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
68
This pie chart gives the composition by weight of municipal solid waste (MSW)before recycling in 2008. 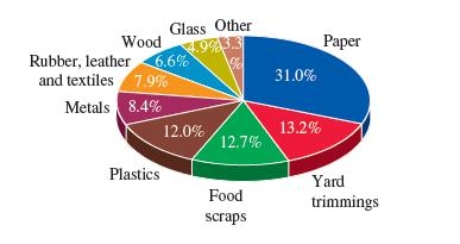
Which of the following statements is not true?
A)There is about 2.6 times more paper waste than plastic waste.
B)Plastic waste is more than the wood and glass waste combined.
C)Plastic waste and food scraps are almost the same.
D)The plastic and paper waste combined account for more than half the MSW.

Which of the following statements is not true?
A)There is about 2.6 times more paper waste than plastic waste.
B)Plastic waste is more than the wood and glass waste combined.
C)Plastic waste and food scraps are almost the same.
D)The plastic and paper waste combined account for more than half the MSW.

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
69
This graph shows the number of PET bottles that are not recycled,by year. 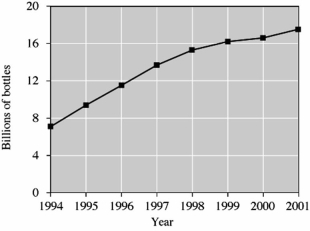
In which year did the number of bottles not recycled exceed 16 billion for the first time?
A)1996
B)1998
C)1999
D)2001

In which year did the number of bottles not recycled exceed 16 billion for the first time?
A)1996
B)1998
C)1999
D)2001

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
70
What is biodegradation?
A)breaking up the plastics into small pieces before putting them into a landfill
B)using bacteria and fungi to "eat" the waste plastic
C)a way to dispose of natural polymers only
D)breaking down polymers into carbon dioxide and water
A)breaking up the plastics into small pieces before putting them into a landfill
B)using bacteria and fungi to "eat" the waste plastic
C)a way to dispose of natural polymers only
D)breaking down polymers into carbon dioxide and water

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
71
This symbol 
Is an example of the coding symbols found on packaging materials.These symbols let consumers know
A)how the plastic was made.
B)how to recycle the package.
C)if the plastic is dangerous.
D)the intended use of the material.

Is an example of the coding symbols found on packaging materials.These symbols let consumers know
A)how the plastic was made.
B)how to recycle the package.
C)if the plastic is dangerous.
D)the intended use of the material.

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
72
Which synthetic polymer has two forms,"crystal" and "expandable"?
A)PET
B)HDPE
C)LDPE
D)PS
A)PET
B)HDPE
C)LDPE
D)PS

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
73
Polyethylene can be represented by this drawing. 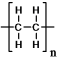
A molecule of polyethylene has n=1000.What is the molar mass of this molecule? The atomic masses are: C = 12.01 g/mol;H = 1.01 g/mol.
A)1000 g/mol
B)14,030 g/mol
C)28,060 g/mol
D)56,120 g/mol

A molecule of polyethylene has n=1000.What is the molar mass of this molecule? The atomic masses are: C = 12.01 g/mol;H = 1.01 g/mol.
A)1000 g/mol
B)14,030 g/mol
C)28,060 g/mol
D)56,120 g/mol

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
74
This symbol 
Is found on the bottom of a Coke bottle.What does the number mean?
A)The bottle may be recycled because it is made from PET.
B)The bottle can not be recycled because it is made from PET.
C)The bottle may be recycled because it is made from HDPE.
D)The bottle can not be recycled because it is made from HDPE.

Is found on the bottom of a Coke bottle.What does the number mean?
A)The bottle may be recycled because it is made from PET.
B)The bottle can not be recycled because it is made from PET.
C)The bottle may be recycled because it is made from HDPE.
D)The bottle can not be recycled because it is made from HDPE.

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
75
These are the densities of some common plastics: PET 1.38 to 1.39 g/mL;HDPE 0.95 to 0.97 g/mL;LDPE 0.90 to 0.91 g/mL.Based on these densities,which plastic/s will sink in an ethanol/water mixture that has a density of 0.94 g/mL?
A)PET only
B)PET and HDPE
C)HDPE only
D)LDPE only
A)PET only
B)PET and HDPE
C)HDPE only
D)LDPE only

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
76
Which type of synthetic polymer is used to make gloves that serve as a barrier to HIV/AIDS?
A)HDPE
B)LDPE
C)PVC
D)PP
A)HDPE
B)LDPE
C)PVC
D)PP

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
77
Consider this table. 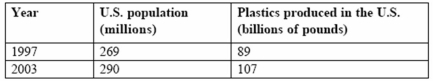
How many pounds of plastics were produced per person in 2003?
A)0.369
B)3.69
C)36.9
D)369

How many pounds of plastics were produced per person in 2003?
A)0.369
B)3.69
C)36.9
D)369

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
78
The regions of a polymer where the long polymer molecules are arranged neatly and tightly in a regular pattern is called the ________ region.
A)crystalline
B)amorphous
C)condensed
D)porous
A)crystalline
B)amorphous
C)condensed
D)porous

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
79
The EPA has determined a solid waste management hierarchy.Which is the most preferred method?
A)incineration and land filling
B)recycling and composting
C)source reduction and reuse
D)condensation
A)incineration and land filling
B)recycling and composting
C)source reduction and reuse
D)condensation

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck
80
One approach to encourage more recycling is to institute a "bottle bill." What is a bottle bill?
A)a bill that outlaws the use of plastic bottles
B)a "container deposit law" that requires customers to pay a deposit for the bottle
C)a large container for recycling bottles
D)a bill that requires equal use of plastic and glass in bottles
A)a bill that outlaws the use of plastic bottles
B)a "container deposit law" that requires customers to pay a deposit for the bottle
C)a large container for recycling bottles
D)a bill that requires equal use of plastic and glass in bottles

Unlock Deck
Unlock for access to all 84 flashcards in this deck.
Unlock Deck
k this deck



