Deck 6: Molecular Shape
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question

Unlock Deck
Sign up to unlock the cards in this deck!
Unlock Deck
Unlock Deck
1/71
Play
Full screen (f)
Deck 6: Molecular Shape
1
How many lone pairs of electrons need to be added to complete this Lewis structure? 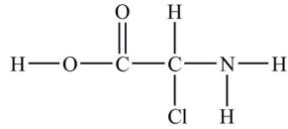
A)5
B)8
C)6
D)1
E)16

A)5
B)8
C)6
D)1
E)16
8
2
In which one of the following species is the central atom (the first atom in the formula) likely to violate the octet rule?
A)BF4−
B)XeO3
C)SiCl4
D)NH3
E)CH2Cl2
A)BF4−
B)XeO3
C)SiCl4
D)NH3
E)CH2Cl2
XeO3
3
What does the abbreviation VSEPR stand for?
A)Very Specific Electron and Proton Repair
B)Variable Selective of Electron and Protons
C)Valence Shell for Every Proton
D)Very Selective Electron Pair theory
E)Valence-Shell Electron-Pair Repulsion
A)Very Specific Electron and Proton Repair
B)Variable Selective of Electron and Protons
C)Valence Shell for Every Proton
D)Very Selective Electron Pair theory
E)Valence-Shell Electron-Pair Repulsion
Valence-Shell Electron-Pair Repulsion
4
The number of resonance structures for the nitrate ion that satisfy the octet rule is
A)1.
B)2.
C)3.
D)4.
E)none of these
A)1.
B)2.
C)3.
D)4.
E)none of these

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
5
How many lone pairs of electrons need to be added to complete this Lewis structure? 
A)5
B)10
C)0
D)1
E)6

A)5
B)10
C)0
D)1
E)6

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
6
Select the correct Lewis structure for NOCl, a reactive material used as an ionizing solvent.
A)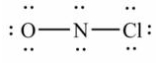
B)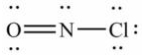
C)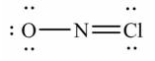
D)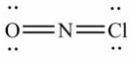
E)None of these choices is correct.
A)
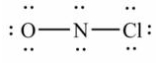
B)
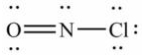
C)
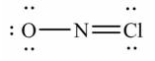
D)
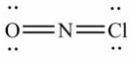
E)None of these choices is correct.

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
7
According to the VSEPR model, the predicted molecular geometry of the SO3 molecule is
A)pyramidal.
B)tetrahedral.
C)trigonal planar.
D)seesaw.
E)square planar.
A)pyramidal.
B)tetrahedral.
C)trigonal planar.
D)seesaw.
E)square planar.

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
8
Select the correct Lewis structure for nitrogen trifluoride, NF3.
A)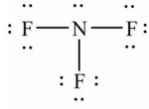
B)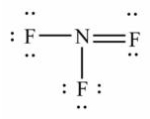
C)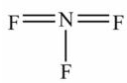
D)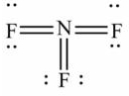
E)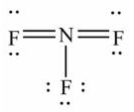
A)
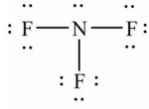
B)

C)
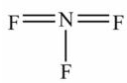
D)
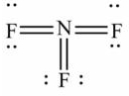
E)
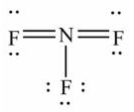

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
9
What is the predicted molecular geometry of the CH4 molecule according to the VSEPR model?
A)tetrahedral
B)trigonal pyramidal
C)trigonal planar
D)square planar
E)seesaw
A)tetrahedral
B)trigonal pyramidal
C)trigonal planar
D)square planar
E)seesaw

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
10
How many total resonance structures can be drawn for the sulfate ion, SO42−?
A)2
B)3
C)4
D)5
E)6
A)2
B)3
C)4
D)5
E)6

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
11
Which of the following is required for the determination of the VSEPR model and the molecular shape?
A)Atomic mass
B)Number of protons
C)Oxidation number
D)Lewis structure
E)None of the answers is correct.
A)Atomic mass
B)Number of protons
C)Oxidation number
D)Lewis structure
E)None of the answers is correct.

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
12
The total number of lone pairs in NCl3 is
A)6.
B)8.
C)9.
D)10.
E)13.
A)6.
B)8.
C)9.
D)10.
E)13.

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
13
The Lewis structure for CS2 is:
A)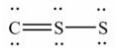
B)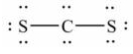
C)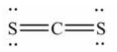
D)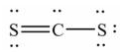
A)
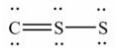
B)

C)

D)
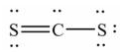

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
14
The total number of bonding electrons in a molecule of formaldehyde (H2CO) is
A)3.
B)4.
C)6.
D)8.
E)18.
A)3.
B)4.
C)6.
D)8.
E)18.

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
15
In which one of the following is the best Lewis structure a resonance structure?
A)SO3
B)BF3
C)I3−
D)SCO (C = central atom)
E)SO32−
A)SO3
B)BF3
C)I3−
D)SCO (C = central atom)
E)SO32−

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
16
The number of resonance structures for the sulfur dioxide molecule that satisfy the octet rule is
A)1.
B)2.
C)3.
D)4.
E)none of these
A)1.
B)2.
C)3.
D)4.
E)none of these

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
17
The Lewis structure for a chlorate ion, ClO3-, should show ____ single bond(s), ____ double bond(s), and ____ lone pair(s).
A)2, 1, 10
B)3, 0, 9
C)2, 1, 8
D)3, 0, 10
E)2, 1, 9
A)2, 1, 10
B)3, 0, 9
C)2, 1, 8
D)3, 0, 10
E)2, 1, 9

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
18
What is wrong with this Lewis structure? 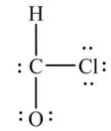
A)There are too many electrons.
B)There are too few electrons.
C)The O atom does not have an octet.
D)The C atom does not have an octet.
E)There is nothing wrong with this Lewis structure.

A)There are too many electrons.
B)There are too few electrons.
C)The O atom does not have an octet.
D)The C atom does not have an octet.
E)There is nothing wrong with this Lewis structure.

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
19
Which of these choices is the best Lewis structure for ozone, O3?
A)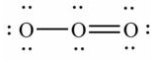
B)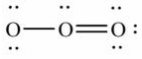
C)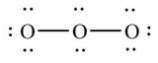
D)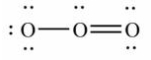
E)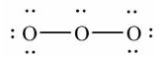
A)

B)
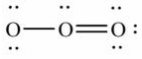
C)

D)
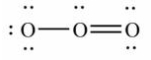
E)


Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
20
Which pair of Lewis structures is not a pair of resonance structures?
A)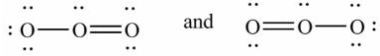
B)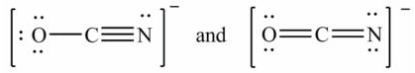
C)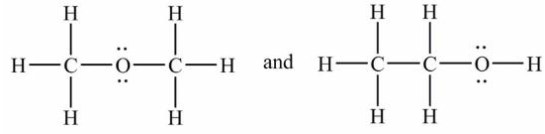
D)All of these pairs are resonance structures of the same species.
E)None of these pairs is resonance structures of the same species.
A)
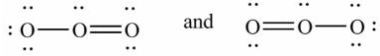
B)
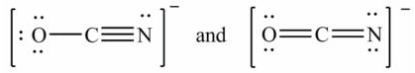
C)

D)All of these pairs are resonance structures of the same species.
E)None of these pairs is resonance structures of the same species.

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
21
Using the VSEPR model, predict the molecular geometry around the central atom in SO32−.
A)trigonal planar
B)trigonal pyramidal
C)tetrahedral
D)trigonal bipyramidal
E)octahedral
A)trigonal planar
B)trigonal pyramidal
C)tetrahedral
D)trigonal bipyramidal
E)octahedral

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
22
What is the molecular geometry of HOF as predicted by the VSEPR model?
A)trigonal pyramidal
B)bent
C)tetrahedral
D)linear
E)trigonal planar
A)trigonal pyramidal
B)bent
C)tetrahedral
D)linear
E)trigonal planar

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
23
According to the VSEPR model, the predicted molecular geometry of SiCl4 is
A)linear.
B)trigonal planar.
C)bent.
D)tetrahedral.
E)trigonal pyramidal.
A)linear.
B)trigonal planar.
C)bent.
D)tetrahedral.
E)trigonal pyramidal.

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
24
Using the VSEPR model, predict the molecular geometry around the central atom in PO43−.
A)trigonal planar
B)trigonal pyramidal
C)tetrahedral
D)trigonal bipyramidal
E)octahedral
A)trigonal planar
B)trigonal pyramidal
C)tetrahedral
D)trigonal bipyramidal
E)octahedral

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
25
What is the number of lone electron pairs on the central atom of a molecule having a linear molecular geometry, such as ClF2−?
A)1
B)2
C)3
D)0
E)4
A)1
B)2
C)3
D)0
E)4

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
26
What is the molecular geometry of NO2− as predicted by the VSEPR model?
A)linear
B)trigonal planar
C)bent
D)tetrahedral
E)trigonal pyramidal
A)linear
B)trigonal planar
C)bent
D)tetrahedral
E)trigonal pyramidal

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
27
What is the molecular geometry of BeH2 as predicted by the VSEPR model?
A)tetrahedral
B)bent
C)trigonal planar
D)linear
E)trigonal pyramidal
A)tetrahedral
B)bent
C)trigonal planar
D)linear
E)trigonal pyramidal

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
28
What is the molecular geometry of N2O as predicted by the VSEPR model? 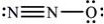
A)trigonal pyramidal
B)trigonal planar
C)tetrahedral
D)bent
E)linear
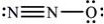
A)trigonal pyramidal
B)trigonal planar
C)tetrahedral
D)bent
E)linear

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
29
According to the VSEPR model, a molecule with the general formula AB2 with two lone pairs on the central atom will have a _____ molecular geometry.
A)linear
B)bent
C)trigonal planar
D)tetrahedral
E)seesaw
A)linear
B)bent
C)trigonal planar
D)tetrahedral
E)seesaw

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
30
According to the VSEPR model, which molecule is predicted to be linear?
A)H2S
B)HCN
C)BF3
D)H2CO
E)SO3
A)H2S
B)HCN
C)BF3
D)H2CO
E)SO3

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
31
According to the VSEPR model, the predicted molecular geometry of ammonia, NH3, is
A)linear.
B)trigonal planar.
C)bent.
D)tetrahedral.
E)trigonal pyramidal.
A)linear.
B)trigonal planar.
C)bent.
D)tetrahedral.
E)trigonal pyramidal.

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
32
What is the number of lone electron pairs on the central atom of a molecule having a trigonal pyramidal molecular geometry, such as NH3?
A)1
B)2
C)3
D)0
E)4
A)1
B)2
C)3
D)0
E)4

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
33
Using the VSEPR model, what is the predicted molecular geometry of the PCl3 molecule?
A)linear
B)bent
C)trigonal planar
D)trigonal pyramidal
E)tetrahedral
A)linear
B)bent
C)trigonal planar
D)trigonal pyramidal
E)tetrahedral

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
34
What is the number of lone electron pairs on the central atom of a molecule having a linear molecular geometry, such as CO2?
A)1
B)2
C)3
D)0
E)4
A)1
B)2
C)3
D)0
E)4

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
35
According to the VSEPR model, a molecule with the general formula AB3 with no lone pairs on the central atom will have a ______ molecular shape.
A)linear
B)bent
C)trigonal planar
D)tetrahedral
E)trigonal pyramidal
A)linear
B)bent
C)trigonal planar
D)tetrahedral
E)trigonal pyramidal

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
36
What is the predicted molecular geometry of the H2O molecule according to the VSEPR model?
A)tetrahedral
B)trigonal pyramidal
C)bent
D)square planar
E)seesaw
A)tetrahedral
B)trigonal pyramidal
C)bent
D)square planar
E)seesaw

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
37
What is the molecular geometry of NOCl as predicted by the VSEPR model? 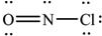
A)linear
B)trigonal planar
C)bent
D)tetrahedral
E)trigonal pyramidal
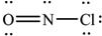
A)linear
B)trigonal planar
C)bent
D)tetrahedral
E)trigonal pyramidal

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
38
What is the molecular geometry of ClF2− as predicted by the VSEPR model?
A)linear
B)bent
C)seesaw
D)T-shaped
E)trigonal bipyramidal
A)linear
B)bent
C)seesaw
D)T-shaped
E)trigonal bipyramidal

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
39
According to the VSEPR model, the molecular geometry of the carbonate ion, CO32−, is
A)square planar.
B)tetrahedral.
C)pyramidal.
D)trigonal planar.
E)octahedral.
A)square planar.
B)tetrahedral.
C)pyramidal.
D)trigonal planar.
E)octahedral.

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
40
What is the molecular geometry of the thiocyanate anion, SCN−, as predicted by the VSEPR model? (Carbon is the central atom.)
A)linear
B)bent
C)tetrahedral
D)trigonal planar
E)trigonal pyramidal
A)linear
B)bent
C)tetrahedral
D)trigonal planar
E)trigonal pyramidal

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
41
Which one of the following pure substances will exhibit hydrogen bonding?
A)CH4
B)H2C=CH2
C)H2O
D)CHCl3
E)CHF3
A)CH4
B)H2C=CH2
C)H2O
D)CHCl3
E)CHF3

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
42
What is the predicted O-C-O bond angle in CO2?
A)60°
B)90°
C)109.5°
D)120°
E)180°
A)60°
B)90°
C)109.5°
D)120°
E)180°

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
43
Which of the following atoms does not participate in hydrogen bonding?
A)S
B)O
C)F
D)N
E)H
A)S
B)O
C)F
D)N
E)H

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
44
Place the following in order of decreasing electronegativity. Mg Cl F
A)Mg > Cl > F
B)F > Cl > Mg
C)Cl > F > Mg
D)Cl > Mg > F
E)F > Mg > Cl
A)Mg > Cl > F
B)F > Cl > Mg
C)Cl > F > Mg
D)Cl > Mg > F
E)F > Mg > Cl

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
45
Which substance will exhibit hydrogen bonding between molecules?
A)(CH3)3N
B)CH3-O-CH3
C)CH3CH2-OH
D)CH3CH2-F
E)HI
A)(CH3)3N
B)CH3-O-CH3
C)CH3CH2-OH
D)CH3CH2-F
E)HI

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
46
Place the following in order of increasing electronegativity. N As K Ca
A)K < Ca < N < As
B)N < As < Ca < K
C)As < Ca < K < N
D)N < K < Ca < As
E)K < Ca < As < N
A)K < Ca < N < As
B)N < As < Ca < K
C)As < Ca < K < N
D)N < K < Ca < As
E)K < Ca < As < N

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
47
The strongest intermolecular interactions between ethyl alcohol (CH3CH2OH) molecules arise from
A)dipole-dipole forces.
B)London dispersion forces.
C)hydrogen bonding.
D)ion-dipole interactions.
E)carbon-oxygen bonds.
A)dipole-dipole forces.
B)London dispersion forces.
C)hydrogen bonding.
D)ion-dipole interactions.
E)carbon-oxygen bonds.

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
48
Which is the most reasonable prediction for the Cl-N-Cl bond angle in NCl3?
A)120°
B)111°
C)109.5°
D)107°
E)90°
A)120°
B)111°
C)109.5°
D)107°
E)90°

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
49
Which of the following is defined as the attractive forces between polar molecules? I.dispersion forces
II)dipole-dipole interactions
Ion-dipole interactions
A)I and III
B)II only
C)III only
D)I, II, and III
E)I only
II)dipole-dipole interactions
Ion-dipole interactions
A)I and III
B)II only
C)III only
D)I, II, and III
E)I only

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
50
Predict the molecular geometry and polarity of the SO2 molecule.
A)linear, polar
B)linear, nonpolar
C)bent, polar
D)bent, nonpolar
E)trigonal planar, polar
A)linear, polar
B)linear, nonpolar
C)bent, polar
D)bent, nonpolar
E)trigonal planar, polar

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
51
Predict the molecular geometry and polarity of the CS2 molecule.
A)linear, polar
B)linear, nonpolar
C)tetrahedral, nonpolar
D)bent, nonpolar
E)bent, polar
A)linear, polar
B)linear, nonpolar
C)tetrahedral, nonpolar
D)bent, nonpolar
E)bent, polar

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
52
Which is the most reasonable prediction for the H-N-H bond angle in NH3?
A)90°
B)109.5°
C)120°
D)107°
E)105°
A)90°
B)109.5°
C)120°
D)107°
E)105°

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
53
According to the VSEPR model, what is the predicted Cl-C-Cl bond angle in CCl4?
A)90°
B)109.5°
C)120°
D)145°
E)180°
A)90°
B)109.5°
C)120°
D)145°
E)180°

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
54
Place the following in order of increasing electronegativity. Se K Cl
A)Cl < Se < K
B)K < Se < Cl
C)K < Cl < Se
D)Se < Cl < K
E)Cl < K < Se
A)Cl < Se < K
B)K < Se < Cl
C)K < Cl < Se
D)Se < Cl < K
E)Cl < K < Se

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
55
Which is the most reasonable prediction for the H-O-H bond angle in H2O?
A)90°
B)109.5°
C)120°
D)107°
E)105°
A)90°
B)109.5°
C)120°
D)107°
E)105°

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
56
Place the following in order of increasing electronegativity. Cs O S
A)O < S < Cs
B)S < O < Cs
C)Cs < S < O
D)Cs < O < S
E)O < Cs < S
A)O < S < Cs
B)S < O < Cs
C)Cs < S < O
D)Cs < O < S
E)O < Cs < S

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
57
Which one of the following molecules is polar?
A)CH4
B)CHBr3
C)F2
D)CBr4
E)CO2
A)CH4
B)CHBr3
C)F2
D)CBr4
E)CO2

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
58
Which substance should exhibit hydrogen bonding in the liquid phase?
A)PH3
B)He
C)H2S
D)CH4
E)CH3OH
A)PH3
B)He
C)H2S
D)CH4
E)CH3OH

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
59
Which is the most reasonable prediction for the H-C-H bond angle in CH4?
A)90°
B)109.5°
C)120°
D)107°
E)105°
A)90°
B)109.5°
C)120°
D)107°
E)105°

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
60
Which one of the following molecules is nonpolar?
A)NH3
B)OF2
C)CH3Cl
D)H2O
E)BeCl2
A)NH3
B)OF2
C)CH3Cl
D)H2O
E)BeCl2

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
61
In which of the following compounds will the molecules not form hydrogen bonds with each other? 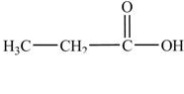
A)
B)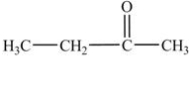
C)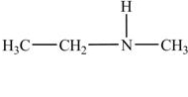
D)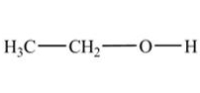

A)
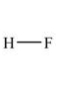
B)

C)

D)


Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
62
Ammonia's unusually high melting point is the result of
A)dipole-dipole forces.
B)London dispersion forces.
C)hydrogen bonding.
D)covalent bonding.
E)ionic bonding.
A)dipole-dipole forces.
B)London dispersion forces.
C)hydrogen bonding.
D)covalent bonding.
E)ionic bonding.

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
63
Helium atoms do not combine to form He2 molecules, yet He atoms do attract one another weakly through
A)dipole-dipole forces.
B)ion-dipole forces.
C)dispersion forces.
D)dipole-induced dipole forces.
E)hydrogen bonding.
A)dipole-dipole forces.
B)ion-dipole forces.
C)dispersion forces.
D)dipole-induced dipole forces.
E)hydrogen bonding.

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
64
Which kinds of intermolecular forces exist between propane molecules? I.dispersion forces
II)dipole-dipole interactions
Ion-dipole interactions
A)I only
B)II only
C)III only
D)I, II, and III
E)I and III
II)dipole-dipole interactions
Ion-dipole interactions
A)I only
B)II only
C)III only
D)I, II, and III
E)I and III

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
65
At a temperature of 27 K, neon condenses due to
A)dipole-dipole forces.
B)London dispersion forces.
C)hydrogen bonding.
D)covalent bonding.
E)intramolecular forces.
A)dipole-dipole forces.
B)London dispersion forces.
C)hydrogen bonding.
D)covalent bonding.
E)intramolecular forces.

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
66
Octane is a component of fuel used in internal combustion engines.The dominant intermolecular forces in octane are
A)dipole-dipole forces.
B)London dispersion forces.
C)hydrogen bonding.
D)covalent bonds.
E)carbon-hydrogen bonds.
A)dipole-dipole forces.
B)London dispersion forces.
C)hydrogen bonding.
D)covalent bonds.
E)carbon-hydrogen bonds.

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
67
The strongest intermolecular interactions between pentane (C5H12) molecules arise from
A)dipole-dipole forces.
B)London dispersion forces.
C)hydrogen bonding.
D)ion-dipole interactions.
E)carbon-carbon bonds.
A)dipole-dipole forces.
B)London dispersion forces.
C)hydrogen bonding.
D)ion-dipole interactions.
E)carbon-carbon bonds.

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
68
Which of the following pure substances has the strongest dispersion forces?
A)C4H10
B)C5H12
C)C6H14
D)C7H16
E)C8H18
A)C4H10
B)C5H12
C)C6H14
D)C7H16
E)C8H18

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
69
Which one of the following pure substance has both dispersion forces and dipole-dipole forces?
A)HCl
B)BCl3
C)Br2
D)H2
E)CO2
A)HCl
B)BCl3
C)Br2
D)H2
E)CO2

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
70
For which of the following pure substances are the intermolecular interactions entirely due to dispersion forces?
A)C2H6
B)CH3OCH3
C)NO2
D)H2S
E)Ca(NO3)2
A)C2H6
B)CH3OCH3
C)NO2
D)H2S
E)Ca(NO3)2

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck
71
The strongest intermolecular interactions between hydrogen fluoride (HF) molecules arise from
A)dipole-dipole forces.
B)London dispersion forces.
C)hydrogen bonding.
D)ion-dipole interactions.
E)ionic bonds.
A)dipole-dipole forces.
B)London dispersion forces.
C)hydrogen bonding.
D)ion-dipole interactions.
E)ionic bonds.

Unlock Deck
Unlock for access to all 71 flashcards in this deck.
Unlock Deck
k this deck



