Deck 2: Electrons and the Periodic Table
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question

Unlock Deck
Sign up to unlock the cards in this deck!
Unlock Deck
Unlock Deck
1/107
Play
Full screen (f)
Deck 2: Electrons and the Periodic Table
1
The size of an atomic orbital is associated with
A)the principal quantum number (n).
B)the angular momentum quantum number (l).
C)the magnetic quantum number (ml).
D)the spin quantum number (ms).
E)the angular momentum and magnetic quantum numbers, together.
A)the principal quantum number (n).
B)the angular momentum quantum number (l).
C)the magnetic quantum number (ml).
D)the spin quantum number (ms).
E)the angular momentum and magnetic quantum numbers, together.
the principal quantum number (n).
2
Determine which sublevel designation is legitimate.
A)1p
B)2p
C)3f
D)4z
A)1p
B)2p
C)3f
D)4z
2p
3
Determine which sublevel designation is not legitimate.
A)1p
B)2s
C)3d
D)4p
A)1p
B)2s
C)3d
D)4p
2s
4
Which of the following electron transitions would be expected to emit any light in the Bohr model of the atom?
A)n = 1 to n = 3
B)n = 5 to n = 6
C)n = 2 to n = 5
D)n = 4 to n = 3
A)n = 1 to n = 3
B)n = 5 to n = 6
C)n = 2 to n = 5
D)n = 4 to n = 3

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
5
Select the arrangement of electromagnetic radiation which starts with the shortest wavelength and increases to longest wavelength.
A)radio, infrared, ultraviolet, gamma rays
B)radio, ultraviolet, infrared, gamma rays
C)gamma rays, radio, ultraviolet, infrared
D)gamma rays, infrared, radio, ultraviolet
E)gamma rays, ultraviolet, infrared, radio
A)radio, infrared, ultraviolet, gamma rays
B)radio, ultraviolet, infrared, gamma rays
C)gamma rays, radio, ultraviolet, infrared
D)gamma rays, infrared, radio, ultraviolet
E)gamma rays, ultraviolet, infrared, radio

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
6
What is the emission of light at only specific wavelengths?
A)Emission spectra
B)Hydrogen spectrum
C)Wave spectra
D)Limited spectra
E)Line spectra
A)Emission spectra
B)Hydrogen spectrum
C)Wave spectra
D)Limited spectra
E)Line spectra

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
7
List the following types of radiation from lowest frequency to highest frequency: microwave, X ray, ultraviolet, visible, and infrared
A)microwave < infrared < visible < ultraviolet < X ray
B)X ray < ultraviolet < visible < infrared < microwave
C)visible < ultraviolet < microwave < X ray < infrared
D)infrared < X ray < microwave < ultraviolet < visible
E)infrared < visible < microwave < ultraviolet < X ray
A)microwave < infrared < visible < ultraviolet < X ray
B)X ray < ultraviolet < visible < infrared < microwave
C)visible < ultraviolet < microwave < X ray < infrared
D)infrared < X ray < microwave < ultraviolet < visible
E)infrared < visible < microwave < ultraviolet < X ray

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
8
Which of the following electron transitions would be expected to absorb any light in the Bohr model of the atom?
A)n = 7 to n = 2
B)n = 5 to n = 6
C)n = 1 to n = 3
D)n = 3 to n = 5
A)n = 7 to n = 2
B)n = 5 to n = 6
C)n = 1 to n = 3
D)n = 3 to n = 5

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
9
Visible light, radio waves, microwave radiation, infrared, ultraviolet radiation, X-rays, and gamma rays all constitute the electromagnetic spectrum.Which of the following characteristics do all of these kinds of radiation share?
A)They all have the ability to generate heat in objects.
B)They all have the same frequencies.
C)They are all the transmission of energy in the form of waves.
D)They have equal energies.
E)They have the same electron spin state.
A)They all have the ability to generate heat in objects.
B)They all have the same frequencies.
C)They are all the transmission of energy in the form of waves.
D)They have equal energies.
E)They have the same electron spin state.

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
10
How many orbitals can have the 4s description in a given atom?
A)1
B)2
C)3
D)5
A)1
B)2
C)3
D)5

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
11
How many orbitals can have the 3d description in a given atom?
A)1
B)2
C)3
D)5
A)1
B)2
C)3
D)5

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
12
Determine which sublevel designation is not legitimate.
A)4s
B)2d
C)3s
D)5p
A)4s
B)2d
C)3s
D)5p

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
13
Which of the following electron transitions would be expected to emit any light in the Bohr model of the atom?
A)n = 1 to n = 4
B)n = 3 to n = 1
C)n = 2 to n = 3
D)n = 5 to n = 7
A)n = 1 to n = 4
B)n = 3 to n = 1
C)n = 2 to n = 3
D)n = 5 to n = 7

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
14
Determine which sublevel designation is legitimate.
A)1f
B)2d
C)3c
D)4s
A)1f
B)2d
C)3c
D)4s

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
15
The number of orbitals in a d subshell is
A)1.
B)2.
C)3.
D)5.
E)7.
A)1.
B)2.
C)3.
D)5.
E)7.

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
16
Atomic orbitals developed using quantum mechanics
A)describe regions of space in which one is most likely to find an electron.
B)describe exact paths for electron motion.
C)give a description of the atomic structure which is essentially the same as the Bohr model.
D)allow scientists to calculate an exact volume for the hydrogen atom.
E)are in conflict with the Heisenberg uncertainty principle.
A)describe regions of space in which one is most likely to find an electron.
B)describe exact paths for electron motion.
C)give a description of the atomic structure which is essentially the same as the Bohr model.
D)allow scientists to calculate an exact volume for the hydrogen atom.
E)are in conflict with the Heisenberg uncertainty principle.

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
17
Select the arrangement of electromagnetic radiation which starts with the lowest energy and increases to the greatest energy.
A) radio, infrared, ultraviolet, gamma rays
B) radio, ultraviolet, infrared, gamma rays
C) gamma rays, infrared, radio, ultraviolet
D) gamma rays, ultraviolet, infrared, radio
E) infrared, ultraviolet, radio, gamma rays
A) radio, infrared, ultraviolet, gamma rays
B) radio, ultraviolet, infrared, gamma rays
C) gamma rays, infrared, radio, ultraviolet
D) gamma rays, ultraviolet, infrared, radio
E) infrared, ultraviolet, radio, gamma rays

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
18
How many orbitals can have the 3p description in a given atom?
A)1
B)2
C)3
D)5
A)1
B)2
C)3
D)5

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
19
Which of the following electron transitions would be expected to absorb any light in the Bohr model of the atom?
A)n = 1 to n = 3
B)n = 3 to n = 2
C)n = 4 to n = 2
D)n = 6 to n = 5
A)n = 1 to n = 3
B)n = 3 to n = 2
C)n = 4 to n = 2
D)n = 6 to n = 5

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
20
How many orbitals can have the 4p description in a given atom?
A)1
B)2
C)3
D)4
A)1
B)2
C)3
D)4

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
21
How many orbitals are there in the n = 4 level of the H-atom?
A)4
B)6
C)8
D)16
E)18
A)4
B)6
C)8
D)16
E)18

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
22
The electronic structure 1s22s22p63s23p64s23d8 refers to the ground state of
A)Kr.
B)Ni.
C)Fe.
D)Pd.
E)None of these choices is correct.
A)Kr.
B)Ni.
C)Fe.
D)Pd.
E)None of these choices is correct.

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
23
Which element has the following ground-state electron configuration? [Kr]5s24d105p3
A)Sn
B)Sb
C)Pb
D)Bi
E)Te
A)Sn
B)Sb
C)Pb
D)Bi
E)Te

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
24
The ground-state electron configuration for an atom of indium is
A)[Kr]5s24p64d5
B)[Ar]4s23d104p1
C)[Ar]4s24p63d5
D)[Kr]5s25p64d5
E)[Kr]5s24d105p1
A)[Kr]5s24p64d5
B)[Ar]4s23d104p1
C)[Ar]4s24p63d5
D)[Kr]5s25p64d5
E)[Kr]5s24d105p1

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
25
The electron configuration of a ground-state vanadium atom is
A)[Ar]4s24d3
B)[Ar]4s24p3
C)[Ar]4s23d3
D)[Ar]3d5
E)[Ar]4s23d7
A)[Ar]4s24d3
B)[Ar]4s24p3
C)[Ar]4s23d3
D)[Ar]3d5
E)[Ar]4s23d7

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
26
Which ground-state atom has an electron configuration described by the following orbital diagram? 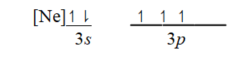
A)Phosphorus
B)nitrogen
C)arsenic
D)vanadium
E)sulfur
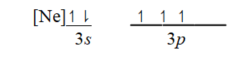
A)Phosphorus
B)nitrogen
C)arsenic
D)vanadium
E)sulfur

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
27
For all atoms of the same element, the 2s orbital is larger than the 1s orbital.

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
28
What is the correct electron configuration for a germanium (Ge) atom?
A)1s22s22p63s23p64s24p2
B)1s22s22p63s23p64s23d104p2
C)1s22s22p63s23p2
D)1s22s23s23p5
E)None of the answers is correct.
A)1s22s22p63s23p64s24p2
B)1s22s22p63s23p64s23d104p2
C)1s22s22p63s23p2
D)1s22s23s23p5
E)None of the answers is correct.

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
29
The orbital diagram for a ground-state carbon atom is 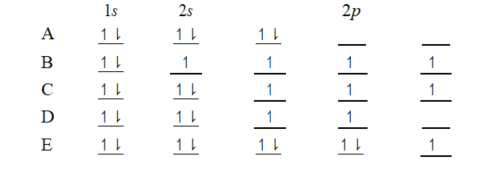


Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
30
The orbital diagram for a ground-state nitrogen atom is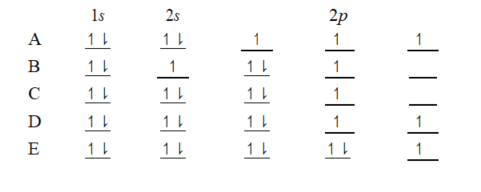


Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
31
Which element has the following ground-state electron configuration? [Kr]5s24d105p2
A)Sn
B)Sb
C)Pb
D)Ge
E)Te
A)Sn
B)Sb
C)Pb
D)Ge
E)Te

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
32
Which element has the following ground-state electron configuration? 1s2 2s2 2p6 3s2
A)Na
B)Mg
C)Al
D)Si
E)Ne
A)Na
B)Mg
C)Al
D)Si
E)Ne

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
33
Which ground-state atom has an electron configuration described by the following orbital diagram? 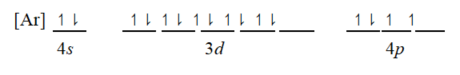
A)phosphorus
B)germanium
C)selenium
D)tellurium
E)potassium
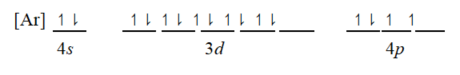
A)phosphorus
B)germanium
C)selenium
D)tellurium
E)potassium

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
34
Select the correct electron configuration for Te (Z = 52).
A)[Kr]5s25p64d8
B)[Kr]5s25d105p4
C)[Kr]5s24d105p6
D)[Kr]5s24f14
E)[Kr]5s24d105p4
A)[Kr]5s25p64d8
B)[Kr]5s25d105p4
C)[Kr]5s24d105p6
D)[Kr]5s24f14
E)[Kr]5s24d105p4

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
35
The ground-state electron configuration of a calcium atom is
A)[Ne]3s2
B)[Ne]3s23p6
C)[Ar]4s13d1
D)[Ar]4s2
E)[Ar]3d2
A)[Ne]3s2
B)[Ne]3s23p6
C)[Ar]4s13d1
D)[Ar]4s2
E)[Ar]3d2

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
36
The orbital diagram for a ground-state oxygen atom is 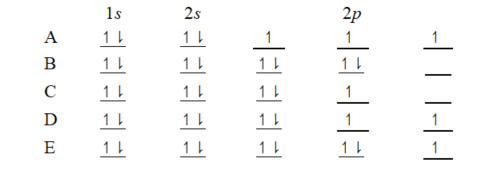


Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
37
How many unpaired electrons does a ground-state atom of sulfur have?
A)0
B)1
C)2
D)3
E)4
A)0
B)1
C)2
D)3
E)4

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
38
The electron configuration of a ground-state Co atom is
A)[Ar]4s23d7
B)1s22s22p63s23d9
C)[Ne]3s23d7
D)[Ar]4s13d5
E)[Ar]4s24d7
A)[Ar]4s23d7
B)1s22s22p63s23d9
C)[Ne]3s23d7
D)[Ar]4s13d5
E)[Ar]4s24d7

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
39
Each shell (principal energy level) of quantum number n contains n subshells.

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
40
Select the correct electron configuration for sulfur (Z = 16).
A)1s21p62s22p6
B)1s22s22p83s23p4
C)1s22s22p83s23p2
D)1s22s22p63s23p4
E)1s22s22p63s23d4
A)1s21p62s22p6
B)1s22s22p83s23p4
C)1s22s22p83s23p2
D)1s22s22p63s23p4
E)1s22s22p63s23d4

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
41
What element is represented by the electron configuration 1s22s22p63s23p64s13d5?
A)Mn
B)Ca
C)K
D)Cr
E)V
A)Mn
B)Ca
C)K
D)Cr
E)V

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
42
In what group of the periodic table is the element with the electron configuration [Ar]4s23d104p3?
A)1A
B)2A
C)3A
D)4A
E)5A
A)1A
B)2A
C)3A
D)4A
E)5A

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
43
How many electrons are in the 4p orbitals of vanadium?
A)0
B)2
C)4
D)5
E)6
A)0
B)2
C)4
D)5
E)6

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
44
How many electrons are there in the 3rd principal energy level (n = 3) of a phosphorus atom?
A)3
B)5
C)6
D)8
E)10
A)3
B)5
C)6
D)8
E)10

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
45
An element with the general electron configuration for its outermost electrons of ns2np1 would be in which element group?
A)2A
B)3A
C)4A
D)5A
E)8A
A)2A
B)3A
C)4A
D)5A
E)8A

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
46
The general electron configuration for noble gas atoms is
A)ns2np6.
B)ns2np5.
C)ns2np4.
D)ns2np3.
E)ns2.
A)ns2np6.
B)ns2np5.
C)ns2np4.
D)ns2np3.
E)ns2.

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
47
How many electrons are in the 4d orbitals of Tc?
A)1
B)2
C)3
D)4
E)5
A)1
B)2
C)3
D)4
E)5

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
48
What is the electron configuration for bromine?
A)1s22s22p63s23p64s14d104p6
B)1s22s22p63s23p64s24d104p5
C)1s22s22p63s23p64s13d104p6
D)1s22s22p63s23p64s23d104p4
E)1s22s22p63s23p64s23d104p5
A)1s22s22p63s23p64s14d104p6
B)1s22s22p63s23p64s24d104p5
C)1s22s22p63s23p64s13d104p6
D)1s22s22p63s23p64s23d104p4
E)1s22s22p63s23p64s23d104p5

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
49
Consider the element with the electron configuration [Kr]5s24d7.This element is
A)a halogen.
B)a transition metal.
C)a nonmetal.
D)an actinide element.
E)a noble gas.
A)a halogen.
B)a transition metal.
C)a nonmetal.
D)an actinide element.
E)a noble gas.

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
50
How many electrons are there in the 2nd principal energy level (n = 2) of a phosphorus atom?
A)3
B)5
C)6
D)8
E)10
A)3
B)5
C)6
D)8
E)10

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
51
What element is represented by the electron configuration 1s22s22p63s23p64s23d104p65s14d10?
A)Ag
B)Rb
C)Cd
D)Sr
E)Cu
A)Ag
B)Rb
C)Cd
D)Sr
E)Cu

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
52
Which of these choices is the general electron configuration for the outermost electrons of elements in the alkaline earth group?
A)ns1
B)ns2
C)ns2np4
D)ns2np
E)ns2np6 (n − 1)d6
A)ns1
B)ns2
C)ns2np4
D)ns2np
E)ns2np6 (n − 1)d6

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
53
What is the electron configuration for silicon?
A)1s22s22p63s13p3
B)1s22s22p63s23p2
C)1s22s22p63s4
D)1s22s22p63p4
E)1s22s22p63s23p3
A)1s22s22p63s13p3
B)1s22s22p63s23p2
C)1s22s22p63s4
D)1s22s22p63p4
E)1s22s22p63s23p3

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
54
The general electron configuration for atoms of the halogen group is
A)ns2np6.
B)ns2np5.
C)ns2np6 (n − 1)d7.
D)ns1.
E)ns2np7.
A)ns2np6.
B)ns2np5.
C)ns2np6 (n − 1)d7.
D)ns1.
E)ns2np7.

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
55
Each of the noble gases has a completely filled p subshell except for which one?
A)Xenon
B)Neon
C)Radon
D)Argon
E)Helium
A)Xenon
B)Neon
C)Radon
D)Argon
E)Helium

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
56
How many electrons are in the 4p orbitals of selenium?
A)0
B)2
C)4
D)5
E)6
A)0
B)2
C)4
D)5
E)6

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
57
Which of the following elements has the largest number of unpaired electrons in the ground state?
A)K
B)V
C)S
D)Si
E)Cl
A)K
B)V
C)S
D)Si
E)Cl

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
58
Consider the element with the electron configuration [Kr]5s24d105p5.This element is
A)a halogen.
B)a transition metal.
C)an alkali metal.
D)an actinide element.
E)a noble gas.
A)a halogen.
B)a transition metal.
C)an alkali metal.
D)an actinide element.
E)a noble gas.

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
59
What is the electron configuration for tungsten?
A)1s22s22p63s23p64s23d104p65s24d105p66s24f145d6
B)1s22s22p63s23p64s23d104p65s24d105p66s14f145d5
C)1s22s22p63s23p64s23d104p65s24d105p66s24f145d4
D)1s22s22p63s23p64s23d104p65s24d105p66s24f145d7
E)1s22s22p63s23p54s23d104p65s24d105p66s24f145d7
A)1s22s22p63s23p64s23d104p65s24d105p66s24f145d6
B)1s22s22p63s23p64s23d104p65s24d105p66s14f145d5
C)1s22s22p63s23p64s23d104p65s24d105p66s24f145d4
D)1s22s22p63s23p64s23d104p65s24d105p66s24f145d7
E)1s22s22p63s23p54s23d104p65s24d105p66s24f145d7

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
60
The general electron configuration for atoms of all elements in Group 5A is
A)ns2np6.
B)ns2np5.
C)ns2np4.
D)ns2np3.
E)ns2np1.
A)ns2np6.
B)ns2np5.
C)ns2np4.
D)ns2np3.
E)ns2np1.

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
61
Which one of these ions has the smallest radius?
A)Cl−
B)K+
C)S2−
D)Na+
E)O2−
A)Cl−
B)K+
C)S2−
D)Na+
E)O2−

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
62
Which of these atoms has the largest radius?
A)B
B)Ga
C)Br
D)Si
E)Cl
A)B
B)Ga
C)Br
D)Si
E)Cl

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
63
Select the element that will gain an electron most easily, based on the periodic trend.
A)Ca
B)Mg
C)O
D)P
A)Ca
B)Mg
C)O
D)P

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
64
Which of the elements listed below has the greatest atomic radius?
A)B
B)Al
C)S
D)P
E)Si
A)B
B)Al
C)S
D)P
E)Si

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
65
An element with the electron configuration [noble gas]ns2(n − 1)d8 has ___________ valence electrons.
A)2
B)6
C)8
D)10
E)None of these choices is correct.
A)2
B)6
C)8
D)10
E)None of these choices is correct.

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
66
Arrange these ions in order of increasing ionic radius: K+ , P3− , S2− , Cl−.
A)K+ < Cl− < S2− < P3−
B)K+ < P3−< S2− < Cl−
C)P3− < S2− < Cl− < K+
D)Cl− < S2− < P3− < K+
E)Cl− < S2− < K+ < P3−
A)K+ < Cl− < S2− < P3−
B)K+ < P3−< S2− < Cl−
C)P3− < S2− < Cl− < K+
D)Cl− < S2− < P3− < K+
E)Cl− < S2− < K+ < P3−

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
67
Select the element that will lose an electron most easily, based on the periodic trend.
A)Na
B)Mg
C)Ar
D)P
A)Na
B)Mg
C)Ar
D)P

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
68
Which of the following elements has the smallest atomic size?
A)Na
B)Ar
C)K
D)Ca
E)Kr
A)Na
B)Ar
C)K
D)Ca
E)Kr

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
69
How many valence electrons does a tin (Sn) atom have?
A)2
B)4
C)14
D)36
E)50
A)2
B)4
C)14
D)36
E)50

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
70
Arrange P, S, and O in order of increasing atomic radius.
A)S < O < P
B)P < S < O
C)O < S < P
D)O < P < S
E)The answer cannot be determined from the data given.
A)S < O < P
B)P < S < O
C)O < S < P
D)O < P < S
E)The answer cannot be determined from the data given.

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
71
Consider the element with the electron configuration [Xe]6s24f7.This element is
A)a halogen.
B)a lanthanide element.
C)a nonmetal.
D)an actinide element.
E)a noble gas.
A)a halogen.
B)a lanthanide element.
C)a nonmetal.
D)an actinide element.
E)a noble gas.

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
72
How many valence electrons does a carbon atom have?
A)1
B)2
C)3
D)4
E)6
A)1
B)2
C)3
D)4
E)6

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
73
Select the element that will lose an electron most easily, based on the periodic trend.
A)Li
B)Na
C)K
D)He
A)Li
B)Na
C)K
D)He

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
74
Which of these elements has the greatest metallic character?
A)Br
B)Se
C)Ni
D)As
E)Si
A)Br
B)Se
C)Ni
D)As
E)Si

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
75
An element with the electron configuration [noble gas]ns2(n − 1)d10np3 has ____________ valence electrons.
A)2
B)3
C)5
D)10
E)15
A)2
B)3
C)5
D)10
E)15

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
76
Which of the following elements has the largest atomic size?
A)S
B)Ca
C)Ba
D)Po
E)Rn
A)S
B)Ca
C)Ba
D)Po
E)Rn

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
77
Select the element that will gain an electron most easily, based on the periodic trend.
A)Rb
B)Al
C)S
D)Na
A)Rb
B)Al
C)S
D)Na

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
78
Which of these elements has the greatest metallic character?
A)Br
B)F
C)Ge
D)Mn
E)Sc
A)Br
B)F
C)Ge
D)Mn
E)Sc

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
79
How does atomic radius change as you move across the periodic table?
A)Atomic radius decreases moving from left to right across a period and increases from top to bottom.
B)Atomic radius increases moving left to right across a period and decreases from top to bottom.
C)Smaller nuclear charge lowers energy; more electrons in an orbital lowers energy.
D)Atomic radius increases diagonally across the periodic table.
E)None of the answers is correct.
A)Atomic radius decreases moving from left to right across a period and increases from top to bottom.
B)Atomic radius increases moving left to right across a period and decreases from top to bottom.
C)Smaller nuclear charge lowers energy; more electrons in an orbital lowers energy.
D)Atomic radius increases diagonally across the periodic table.
E)None of the answers is correct.

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck
80
Which of these atoms has the smallest radius?
A)Al
B)P
C)As
D)Te
E)Na
A)Al
B)P
C)As
D)Te
E)Na

Unlock Deck
Unlock for access to all 107 flashcards in this deck.
Unlock Deck
k this deck



