Deck 1: Atoms and Elements
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question
Question

Unlock Deck
Sign up to unlock the cards in this deck!
Unlock Deck
Unlock Deck
1/50
Play
Full screen (f)
Deck 1: Atoms and Elements
1
Which of the following is a nonmetal?
A)Lithium, Li, Z = 3
B)Bromine, Br, Z = 35
C)Mercury, Hg, Z = 80
D)Bismuth, Bi, Z = 83
E)Sodium, Na, Z = 11
A)Lithium, Li, Z = 3
B)Bromine, Br, Z = 35
C)Mercury, Hg, Z = 80
D)Bismuth, Bi, Z = 83
E)Sodium, Na, Z = 11
Bromine, Br, Z = 35
2
A row of the periodic table is called a(n)
A)group.
B)period.
C)isotopic mixture.
D)family.
E)subshell.
A)group.
B)period.
C)isotopic mixture.
D)family.
E)subshell.
period.
3
Almost all the mass of an atom is concentrated in the nucleus.
True
4
When applying the scientific method, it is important to avoid any form of hypothesis.

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
5
What is the term for the number of protons in the nucleus of each atom of an element? It also indicates the number of electrons in the atom.
A)Isotope number
B)Mass number
C)Mass-to-charge ratio
D)Atomic number
E)Atomic mass units
A)Isotope number
B)Mass number
C)Mass-to-charge ratio
D)Atomic number
E)Atomic mass units

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
6
The elements in a column of the periodic table are known as
A)metalloids.
B)a period.
C)noble gases.
D)a group.
E)nonmetals.
A)metalloids.
B)a period.
C)noble gases.
D)a group.
E)nonmetals.

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
7
Which of the following is a metalloid?
A)Carbon, C, Z = 6
B)Sulfur, S, Z = 16
C)Germanium, Ge, Z = 32
D)Iridium, Ir, Z = 77
E)Bromine, Br, Z = 35
A)Carbon, C, Z = 6
B)Sulfur, S, Z = 16
C)Germanium, Ge, Z = 32
D)Iridium, Ir, Z = 77
E)Bromine, Br, Z = 35

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
8
The mass of a neutron is equal to the mass of a proton plus the mass of an electron.

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
9
Rutherford's experiment with alpha particle scattering by gold foil established that
A)protons are not evenly distributed throughout an atom.
B)electrons have a negative charge.
C)electrons have a positive charge.
D)atoms are made of protons, neutrons, and electrons.
E)protons are 1840 times heavier than electrons.
A)protons are not evenly distributed throughout an atom.
B)electrons have a negative charge.
C)electrons have a positive charge.
D)atoms are made of protons, neutrons, and electrons.
E)protons are 1840 times heavier than electrons.

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
10
Which of these scientists developed the nuclear model of the atom?
A)John Dalton
B)Robert Millikan
C)J.J.Thomson
D)Henry Moseley
E)Ernest Rutherford
A)John Dalton
B)Robert Millikan
C)J.J.Thomson
D)Henry Moseley
E)Ernest Rutherford

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
11
Which one of the following statements about atoms and subatomic particles is correct?
A)Rutherford discovered the atomic nucleus by bombarding gold foil with electrons
B)The proton and the neutron have identical masses.
C)The neutron's mass is equal to that of a proton plus an electron.
D)A neutral atom contains equal numbers of protons and electrons.
E)An atomic nucleus contains equal numbers of protons and neutrons.
A)Rutherford discovered the atomic nucleus by bombarding gold foil with electrons
B)The proton and the neutron have identical masses.
C)The neutron's mass is equal to that of a proton plus an electron.
D)A neutral atom contains equal numbers of protons and electrons.
E)An atomic nucleus contains equal numbers of protons and neutrons.

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
12
Which of the following is a metal?
A)Nitrogen, N, Z = 7
B)Phosphorus, P, Z = 15
C)Arsenic, As, Z = 33
D)Thallium, Tl, Z = 81
E)Silicon, Si, Z = 14
A)Nitrogen, N, Z = 7
B)Phosphorus, P, Z = 15
C)Arsenic, As, Z = 33
D)Thallium, Tl, Z = 81
E)Silicon, Si, Z = 14

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
13
In the periodic table, atoms are arranged in order of
A)increasing atomic mass.
B)increasing atomic number.
C)physical properties.
D)periodicity.
E)chemical reactivities.
A)increasing atomic mass.
B)increasing atomic number.
C)physical properties.
D)periodicity.
E)chemical reactivities.

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
14
Who is credited with discovering the atomic nucleus?
A)Dalton
B)Gay-Lussac
C)Thomson
D)Chadwick
E)Rutherford
A)Dalton
B)Gay-Lussac
C)Thomson
D)Chadwick
E)Rutherford

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
15
Which of the following is an example of an observation?
A)Gases expand as their temperature increases because the gas molecules are moving more rapidly.
B)Paraffin wax begins to melt at 57°C.
C)Three samples of wax are heated to 75°C.
D)The force acting on an object is equal to its mass times its acceleration.
E)Will all waxes melt at the same temperature?
A)Gases expand as their temperature increases because the gas molecules are moving more rapidly.
B)Paraffin wax begins to melt at 57°C.
C)Three samples of wax are heated to 75°C.
D)The force acting on an object is equal to its mass times its acceleration.
E)Will all waxes melt at the same temperature?

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
16
Rutherford bombarded gold foil with alpha (α) particles and found that a small percentage of the particles were deflected.Which of the following was not accounted for by the model he proposed for the structure of the atom?
A)the small size of the nucleus
B)the charge on the nucleus
C)the total mass of the atom
D)the existence of protons
E)the presence of electrons outside the nucleus
A)the small size of the nucleus
B)the charge on the nucleus
C)the total mass of the atom
D)the existence of protons
E)the presence of electrons outside the nucleus

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
17
The elements in Group 7A are known by what name?
A)Transition metals
B)Halogens
C)Alkali metals
D)Alkaline earth metals
E)Noble gases
A)Transition metals
B)Halogens
C)Alkali metals
D)Alkaline earth metals
E)Noble gases

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
18
What is a unifying principle that explains a body of experimental observations?
A)Law
B)Hypothesis
C)Theory
D)Phenomena
E)Prediction
A)Law
B)Hypothesis
C)Theory
D)Phenomena
E)Prediction

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
19
What is the term used for findings that are summarized based on a pattern or trend?
A)Law
B)Hypothesis
C)Theory
D)Phenomena
E)Prediction
A)Law
B)Hypothesis
C)Theory
D)Phenomena
E)Prediction

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
20
The elements in Group 2A are known by what name?
A)Transition metals
B)Halogens
C)Alkali metals
D)Alkaline earth metals
E)Noble gases
A)Transition metals
B)Halogens
C)Alkali metals
D)Alkaline earth metals
E)Noble gases

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
21
Which of these elements exhibits chemical behavior similar to that of potassium?
A)magnesium
B)sodium
C)sulfur
D)chlorine
E)iron
A)magnesium
B)sodium
C)sulfur
D)chlorine
E)iron

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
22
What element is represented by X in the atomic symbol notation ? 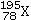
A)Iridium
B)Platinum
C)Palladium
D)Selenium
E)Magnesium
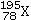
A)Iridium
B)Platinum
C)Palladium
D)Selenium
E)Magnesium

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
23
Which element would be expected to have properties similar to calcium?
A)Ba
B)K
C)Sc
D)Na
E)Rb
A)Ba
B)K
C)Sc
D)Na
E)Rb

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
24
Determine the number of electrons and identify the correct symbol for an atom with 17 protons and 18 neutrons.
A)17 electrons,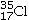
B)18 electrons,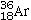
C)17 electrons,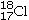
D)17 electrons,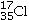
E)18 electrons,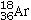
A)17 electrons,
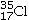
B)18 electrons,
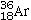
C)17 electrons,
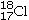
D)17 electrons,
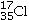
E)18 electrons,
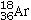

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
25
Which element would be expected to have properties similar to antimony?
A)Se
B)Sn
C)P
D)As
E)Pb
A)Se
B)Sn
C)P
D)As
E)Pb

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
26
Which of these elements exhibits chemical behavior similar to that of silver?
A)nickel
B)gold
C)sulfur
D)chlorine
E)iron
A)nickel
B)gold
C)sulfur
D)chlorine
E)iron

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
27
In what groups are transition metals located?
A)1A, 7A, and 1B
B)2A, 4A, and 7A
C)1B through 8B
D)2B and 3B through 6B
E)3A through 6A
A)1A, 7A, and 1B
B)2A, 4A, and 7A
C)1B through 8B
D)2B and 3B through 6B
E)3A through 6A

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
28
Which one of these elements is a transition element?
A)Sr
B)Pb
C)As
D)Fe
E)H
A)Sr
B)Pb
C)As
D)Fe
E)H

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
29
Determine the number of protons, electrons, and neutrons for the isotope gold-118.The symbol for gold is Au.
A)118 protons, 118 electrons, 79 neutrons
B)79 protons, 79 electrons, 118 neutrons
C)79 protons, 79 electrons, 39 neutrons
D)118 protons, 118 electrons, 39 neutrons
E)79 protons, 39 electrons, 118 neutrons
A)118 protons, 118 electrons, 79 neutrons
B)79 protons, 79 electrons, 118 neutrons
C)79 protons, 79 electrons, 39 neutrons
D)118 protons, 118 electrons, 39 neutrons
E)79 protons, 39 electrons, 118 neutrons

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
30
Which of these elements is chemically similar to magnesium?
A)Sulfur
B)Calcium
C)Iron
D)Nickel
E)Potassium
A)Sulfur
B)Calcium
C)Iron
D)Nickel
E)Potassium

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
31
Lead (Pb) is a main group element.

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
32
Which of these elements exhibits chemical behavior similar to that of oxygen?
A)magnesium
B)sodium
C)sulfur
D)chlorine
E)iron
A)magnesium
B)sodium
C)sulfur
D)chlorine
E)iron

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
33
Copper (Cu) is a transition metal.

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
34
Which of these elements is chemically similar to oxygen?
A)Sulfur
B)Calcium
C)Iron
D)Nickel
E)Potassium
A)Sulfur
B)Calcium
C)Iron
D)Nickel
E)Potassium

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
35
Determine the number of protons and identify the correct symbol for an atom with 20 neutrons and 20 electrons.
A)20 protons,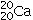
B)20 protons,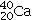
C)20 protons,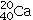
D)40 protons,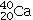
E)40 protons,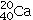
A)20 protons,
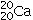
B)20 protons,
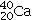
C)20 protons,
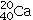
D)40 protons,
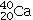
E)40 protons,
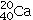

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
36
Which element would be expected to have properties similar to argon?
A)F
B)Cl
C)H
D)Br
E)Kr
A)F
B)Cl
C)H
D)Br
E)Kr

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
37
What elements and groups have properties that are most similar to those of chlorine?
A)F, Br, I, and nonmetals in Group 7A
B)Cl, K, C, and metals in Group 1B
C)N, P, As, and lanthanides
D)He, Ne, Xe, and nonmetals in Group 7A
E)O, S, and P
A)F, Br, I, and nonmetals in Group 7A
B)Cl, K, C, and metals in Group 1B
C)N, P, As, and lanthanides
D)He, Ne, Xe, and nonmetals in Group 7A
E)O, S, and P

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
38
Which one of these elements is a transition element?
A)Nickel
B)Tin
C)Sodium
D)Sulfur
E)Calcium
A)Nickel
B)Tin
C)Sodium
D)Sulfur
E)Calcium

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
39
The alkali metal elements are found in _______ of the periodic table.
A)Group 1A
B)Group 2A
C)Group 3A
D)Period 7
E)Period 1
A)Group 1A
B)Group 2A
C)Group 3A
D)Period 7
E)Period 1

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
40
Which of these elements is chemically similar to potassium?
A)calcium
B)arsenic
C)phosphorus
D)cerium
E)cesium
A)calcium
B)arsenic
C)phosphorus
D)cerium
E)cesium

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
41
Silicon, which makes up about 25% of Earth's crust by mass, is used widely in the modern electronics industry.It has three naturally occurring isotopes, 28Si, 29Si, and 30Si.Calculate the atomic mass of silicon. Isotope Isotopic Mass (amu)
Abudance
28Si
%
27)976927
92)22
29Si
28)976495
4)69
30Si
29)973770
3)09
A)29.2252 amu
B)28.9757 amu
C)28.7260 amu
D)28.0855 amu
E)27.9801 amu
Abudance
28Si
%
27)976927
92)22
29Si
28)976495
4)69
30Si
29)973770
3)09
A)29.2252 amu
B)28.9757 amu
C)28.7260 amu
D)28.0855 amu
E)27.9801 amu

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
42
Which isotope is not possible?
A)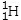
B)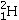
C)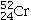
D)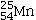
E)All of these isotopes are possible.
A)
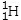
B)
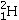
C)
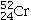
D)
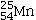
E)All of these isotopes are possible.

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
43
How many neutrons are there in an atom of lead whose mass number is 208?
A)82
B)126
C)208
D)290
E)none of them
A)82
B)126
C)208
D)290
E)none of them

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
44
C(graphite) and C(diamond) are examples of:
A)isotopes of carbon.
B)allotropes of carbon.
C)the law of definite proportions.
D)different carbon ions.
A)isotopes of carbon.
B)allotropes of carbon.
C)the law of definite proportions.
D)different carbon ions.

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
45
Lithium forms compounds which are used in dry cells, storage batteries, and in high-temperature lubricants.It has two naturally occurring isotopes, 6Li (isotopic mass = 6.015123 amu) and 7Li (isotopic mass = 7.016005 amu).Lithium has an atomic mass of 6.9412 amu.What is the percent abundance of lithium-6?
A)92.53%
B)86.65%
C)49.47%
D)7.47%
E)6.015%
A)92.53%
B)86.65%
C)49.47%
D)7.47%
E)6.015%

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
46
Bromine is the only nonmetal that is a liquid at room temperature.Consider the isotope bromine-81, 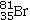 .Select the combination which lists the correct atomic number, number of neutrons, and mass number, respectively.
.Select the combination which lists the correct atomic number, number of neutrons, and mass number, respectively.
A)35, 46, 81
B)35, 81, 46
C)81, 46, 35
D)46, 81, 35
E)35, 81, 116
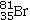 .Select the combination which lists the correct atomic number, number of neutrons, and mass number, respectively.
.Select the combination which lists the correct atomic number, number of neutrons, and mass number, respectively.A)35, 46, 81
B)35, 81, 46
C)81, 46, 35
D)46, 81, 35
E)35, 81, 116

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
47
Two isotopes of an element differ only in their
A)symbol.
B)atomic number.
C)atomic mass.
D)number of protons.
E)number of electrons.
A)symbol.
B)atomic number.
C)atomic mass.
D)number of protons.
E)number of electrons.

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
48
Give the number of protons (p), electrons (e), and neutrons (n) in one atom of chlorine-37.
A)37 p, 37 e, 17 n
B)17 p, 17 e, 37 n
C)17 p, 17 e, 20 n
D)37 p, 17 e, 20 n
E)17 p, 37 e, 17 n
A)37 p, 37 e, 17 n
B)17 p, 17 e, 37 n
C)17 p, 17 e, 20 n
D)37 p, 17 e, 20 n
E)17 p, 37 e, 17 n

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
49
An atom of the isotope sulfur-31 consists of how many protons, neutrons, and electrons? (p = proton, n = neutron, e = electron)
A)15 p, 16 n, 15 e
B)16 p, 15 n, 16 e
C)16 p, 31 n, 16 e
D)32 p, 31 n, 32 e
E)16 p, 16 n, 15 e
A)15 p, 16 n, 15 e
B)16 p, 15 n, 16 e
C)16 p, 31 n, 16 e
D)32 p, 31 n, 32 e
E)16 p, 16 n, 15 e

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck
50
Atoms of the same element with different mass numbers are called
A)ions.
B)neutrons.
C)chemical groups.
D)chemical families.
E)isotopes.
A)ions.
B)neutrons.
C)chemical groups.
D)chemical families.
E)isotopes.

Unlock Deck
Unlock for access to all 50 flashcards in this deck.
Unlock Deck
k this deck



