Multiple Choice
Hexachlorophene is used as a disinfectant in germicidal soaps. What mass of hexachlorophene ( 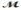 = 406.9 g/mol) must be added to 125 g of chloroform to give a solution with a boiling point of 62.60°C? Kb = 3.63°C/m, boiling point of pure chloroform = 61.70°C
= 406.9 g/mol) must be added to 125 g of chloroform to give a solution with a boiling point of 62.60°C? Kb = 3.63°C/m, boiling point of pure chloroform = 61.70°C
A) 12.6 g
B) 17.2 g
C) 31.0 g
D) 34.4 g
E) 101 g
Correct Answer:
Verified
Related Questions
Q61: Barbiturates are synthetic drugs used as sedatives
Q62: Cinnamaldehyde ( Q64: Calculate the freezing point of a solution Q67: Which one of the following pairs of![]()
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents