The thermochemical equation for the formation of ammonia from elemental nitrogen and hydrogen is as follows. N2(g) + 3 H2(g) 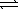 2 NH3(g)
2 NH3(g)
ΔH = -92.2 kJ
Given a system that is initially at equilibrium,which of the following actions cause the reaction to proceed to the left?
A) adding N2(g)
B) removing NH3(g)
C) adding a catalyst
D) decreasing the temperature
E) removing H2(g)
Correct Answer:
Verified
Q57: At 800 K,the equilibrium constant,Kp,for the following
Q58: For the equilibrium PCl5(g) Q59: If Kc = 0.124 for A2 + Q60: Consider the reaction H2 + I2 Q61: In 1913,the Haber-Bosch process was patented.The product Q63: In which of the following reactions does Q64: Consider the following equilibrium. PCl3(g)+ Cl2(g) Q65: Consider the following equilibria. PbBr2(s) Q66: Given the equilibrium constants for the following Q67: If a stress is applied to an![]()

Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents