Sodium metal reacts with water to produce hydrogen gas and sodium hydroxide according to the chemical equation shown below. When 0.0 15 mol of Na is added to 100.00 g of water, the temperature of the resulting solution rises from 25.00 °C to 31.45 °C. If the specific heat of the solution is 4.18 J 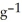
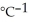 , calculate ΔH for the reaction, as written. 2Na(s) + 2H2O(l) → 2NaOH(aq) + H2(g)
, calculate ΔH for the reaction, as written. 2Na(s) + 2H2O(l) → 2NaOH(aq) + H2(g) 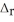 H= ?
H= ?
A) -5.41 kJ
B) -90.0 kJ
C) -180 kJ
D) -360 kJ
Correct Answer:
Verified
Q111: For a particular process that is carried
Q130: Define chemical energy.
Q131: Where does the energy absorbed during an
Q136: Hydrogen peroxide decomposes to water and oxygen
Q138: The specific heat capacity of liquid mercury
Q139: The specific heat capacity of solid copper
Q140: A 50.0 g sample of liquid water
Q142: Match the following.
-energy associated with the relative
Q145: Match the following.
--ΔU
A)energy flows out of the
Q148: Match the following.
--q
A)energy flows out of the
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents