The mole fraction of argon in dry air near sea level is 1.818 × 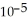 , where the molar mass of argon is 39.948. The concentration of argon in the atmosphere is ________ ppm.
, where the molar mass of argon is 39.948. The concentration of argon in the atmosphere is ________ ppm.
A) 9.3 × 10-9
B) 0.93
C) 9.3 × 106
D) 1.1 × 108
E) 18.18
Correct Answer:
Verified
Q63: Why does hot air rise?
Q148: Consider a container of gas under a
Q149: Why doesn't Dalton's law of partial pressures
Q151: Match the following.
-Avogadro's law
A)PV = nRT
B)V1/T1 =
Q152: What is the temperature of SO2 gas
Q154: Match the following.
-sphygmomanometer
A)PV = nRT
B)V1/T1 = V2/T2
C)V1/n1
Q155: Name the major gas in dry air.
Q156: Match the following.
-Dalton's law
A)PV = nRT
B)V1/T1 =
Q157: Match the following.
-barometer
A)PV = nRT
B)V1/T1 = V2/T2
C)V1/n1
Q158: Match the following.
-Charles's law
A)PV = nRT
B)V1/T1 =
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents