Lead ions can be precipitated from aqueous solutions by the addition of aqueous iodide: 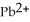 (aq) +
(aq) + 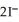 (aq) →
(aq) → 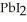 (s) Lead iodide is virtually insoluble in water so that the reaction appears to go to completion. How many millilitres of 3.550 mol L-1 HI(aq) must be added to a solution containing 0.700 mol of
(s) Lead iodide is virtually insoluble in water so that the reaction appears to go to completion. How many millilitres of 3.550 mol L-1 HI(aq) must be added to a solution containing 0.700 mol of 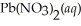 to completely precipitate the lead?
to completely precipitate the lead?
A) 197 mL
B) 0.197 mL
C) 394 mL
D) 0.394 mL
E) 2.54 × 10-3 mL
Correct Answer:
Verified
Q89: Which one of the following compounds is
Q108: Which of the following compounds is an
Q109: What is the reduction half-reaction for the
Q114: What reagent could not be used to
Q117: Which of the following compounds is an
Q131: What reagent could be used to separate
Q155: Which pair of compounds is soluble in
Q156: What is the stoichiometric coefficient for oxygen
Q157: Which of the following compounds is an
Q169: How many H+ ions can the acid,H2SO4,donate
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents