Calcium oxide reacts with water in a combination reaction to produce calcium hydroxide: CaO(s) + 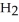 O(l) →
O(l) → 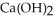 (s)
(s)
In a particular experiment, 5.00 g sample of CaO is reacted with excess water and 6.11 g of 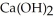 is recovered. What is the percent yield in this experiment?
is recovered. What is the percent yield in this experiment?
A) 81.8
B) 1.22
C) 108
D) 122
E) 92.5
Correct Answer:
Verified
Q102: How many moles of CuO can be
Q107: How many moles of BCl3 are needed
Q121: How many grams of calcium chloride are
Q122: Dinitrogen monoxide gas decomposes to form nitrogen
Q126: When 7.00 × 1022 molecules of ammonia
Q137: If 294 grams of FeS2 is allowed
Q138: When silver nitrate reacts with barium chloride,silver
Q189: Magnesium burns in air with a dazzling
Q193: If the density of ethanol, C2H5OH, is
Q194: Lithium and nitrogen react in a combination
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents