Nitrosyl bromide decomposes according to the following equation. 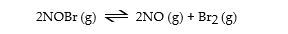
A sample of NOBr (0.64 mol) was placed in a 1.00- L flask containing no NO or Br2. At equilibrium the flask contained 0.46 mol of NOBr. How many moles of NO and Br2, respectively, are in the flask at equilibrium?
A) 0.46, 0.23
B) 0.46, 0.46
C) 0.18, 0.360
D) 0.18, 0.18
E) 0.18, 0.090
Correct Answer:
Verified
Q2: How is the reaction quotient used to
Q3: What role did Karl Bosch play in
Q4: The equilibrium- constant expression depends on the
Q5: The equilibrium- constant expression for the reaction
Q6: At 900 K, the equilibrium constant (Kp)
Q7: The Keq for the equilibrium below is
Q8: The equilibrium constant for reaction 1 is
Q9: The Keq for the equilibrium below is
Q10: The equilibrium constant (Kp) for the reaction
Q11: The equilibrium constant for the gas phase
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents