In this Lewis structure, which atom does NOT have a complete valence? 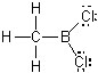
A) H
B) B
C) C
D) Cl
Correct Answer:
Verified
Q3: Which compound has an expanded octet around
Q6: How many atoms in this molecule have
Q7: Which statement is CORRECT concerning the cyanide
Q8: What is the electronic geometry around the
Q9: Which Lewis structure, although acceptable, LEAST describes
Q10: Which molecule does NOT have a lone
Q12: What is the formal charge on the
Q17: A neutral carbon atom has _ valence
Q18: A oxygen atom with one bond and
Q19: A neutral sulfur atom has _ valence
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents