If the absolute temperature of a gas is doubled, what is the change in the average kinetic energy of its molecules?
A) no change
B) increases by a factor of 2
C) decreases by a factor of 2
D) increases by a factor of 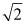
E) decreases by a factor of 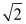
Correct Answer:
Verified
Q31: A cylinder of volume 50 L contains
Q42: At room temperature,which of the following diatomic
Q45: The oxygen (molar mass = 32 g/mol)and
Q46: A room measures 3 m × 4
Q48: At what common Celsius temperature is the
Q50: Two monoatomic gases, helium and neon, are
Q51: One mole of hydrogen gas molar mass
Q52: If the absolute temperature of a gas
Q54: Two monoatomic gases, helium and neon, are
Q55: Doubling the Kelvin temperature of a gas
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents