On the basis of the kinetic theory of gases, when the absolute temperature is doubled, the average kinetic energy of the gas molecules changes by a factor of
A) 16.
B) 2.
C) 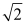 .
.
D) 4.
E) 0.5.
Correct Answer:
Verified
Q67: When the temperature of an ideal gas
Q67: If α is the coefficient of linear
Q68: A room measures 3 m × 4
Q69: If the rms speed of oxygen molecules
Q70: The coefficient of linear expansion for most
Q72: Five molecules of a gas have the
Q74: The coefficient of thermal expansion of water
Q74: If the rms speed of nitrogen molecules
Q75: The coefficient of linear expansion for a
Q76: A volume of an ideal gas goes
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents