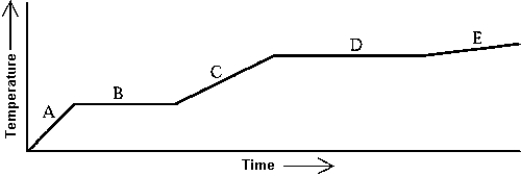
 Heat is added to a substance at a constant rate. The substance starts as a solid and is melted; the liquid is heated and vaporized; finally, the vapor is heated at constant volume. This process is shown in the graph. The specific heat at constant volume of the vapor can be found by
Heat is added to a substance at a constant rate. The substance starts as a solid and is melted; the liquid is heated and vaporized; finally, the vapor is heated at constant volume. This process is shown in the graph. The specific heat at constant volume of the vapor can be found by
A) multiplying the length of B in seconds) by the rate at which heat is added, and dividing by the mass of the substance.
B) multiplying the length of D in seconds) by the rate at which heat is added, and dividing by the mass of the substance.
C) dividing the rate at which heat is added by the product of the slope of A and the mass of the substance.
D) dividing the rate at which heat is added by the product of the slope of C and the mass of the substance.
E) dividing the rate at which heat is added by the product of the slope of E and the mass of the substance.
Correct Answer:
Verified
Q74: If the rms speed of nitrogen molecules
Q78: The rms speed of oxygen molecules is
Q79: Q80: Modern railway tracks consist of continuous welded-steel Q81: Which of the following statements about heat Q83: The quantity of heat absorbed by a![]()
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents