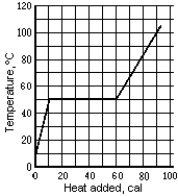
 The graph shows the temperature of a 1.0-g sample of material as heat is added to it. The material is initially a solid at 10oC. The pressure remains constant, and there is no chemical change. The specific heat of the liquid phase is
The graph shows the temperature of a 1.0-g sample of material as heat is added to it. The material is initially a solid at 10oC. The pressure remains constant, and there is no chemical change. The specific heat of the liquid phase is
A) 0.84 cal/g · Co.
B) 0.25 cal/g · Co.
C) 1.6 cal/g · Co.
D) 1.7 cal/g · Co.
E) None of these is correct.
Correct Answer:
Verified
Q29: Which of the following processes of heat
Q37: If the absolute temperature of the filament
Q41: The main process by which heat is
Q45: If the absolute temperature of an object
Q46: If the absolute temperature of an object
Q131: If the thickness of a uniform wall
Q134: If the thickness of a uniform wall
Q135: Earth receives approximately 1.7 × 1017 J
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents