According to the balanced chemical equation below, if, you react 28 grams of butene with excess hydrogen, how many grams of butane would you expect to get? 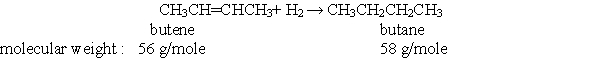
A) 0.0 g
B) 29 g
C) 58 g
D) 112 g
E) 116 g
Correct Answer:
Verified
Q39: How many atoms of hydrogen are present
Q53: Is this chemical equation balanced? Mg(OH)2 +
Q56: The balanced chemical equation for the combustion
Q65: Pentane (C5H12) reacts with oxygen gas (O2)
Q68: Pentane (C5H12) reacts with oxygen gas (O2)
Q69: The name for the heat energy released
Q72: Pentane (C5H12) reacts with oxygen gas (O2)
Q73: Acetylene (C2H2) is a small organic molecule
Q74: According to the balanced chemical equation below,
Q75: Which of the following biological molecules are
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents