Consider the figure that shows ΔG° for a chemical process plotted against absolute temperature. 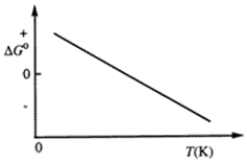 Which one of the following is an incorrect conclusion, based on the information in the diagram?
Which one of the following is an incorrect conclusion, based on the information in the diagram?
A) Δ H° > 0
B) Δ S° > 0
C) The reaction is spontaneous at high temperatures.
D) Δ S° increases with temperature while Δ H° remains constant.
E) There exists a certain temperature at which Δ H° = TΔ S°.
Correct Answer:
Verified
Q60: Given: H2O(l) → H2O(g) ΔH° = 40.7
Q61: "A diamond is forever" is one of
Q62: Use the thermodynamic data at 298 K
Q63: Use the given data at 298 K
Q64: What is the free energy change, ΔG°,
Q66: The temperature at which the following process
Q67: Sulfuryl dichloride is formed when sulfur dioxide
Q68: Iron(III) oxide can be reduced by carbon
Q69: Hydrogen sulfide decomposes according to the following
Q70: The temperature at which the following process
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents