Identify the correct balanced chemical equation for the cell: 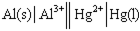
A) Al( s ) + Hg2+( aq ) → Al3+( aq ) + Hg( l )
B) 2 Al( s ) + Hg2+( aq ) → 2 Al3+( aq ) + Hg( l )
C) Al( s ) + 3 Hg2+( aq ) → Al3+( aq ) + 3 Hg( l )
D) 2 Al( s ) + 3 Hg2+( aq ) → 2 Al3+( aq ) + 3 Hg( l )
Correct Answer:
Verified
Q30: Given a Galvanic cell: Q31: What is the E value for the Q32: An oxidation occurs at the _ of Q33: The salt bridge between 1/2 reactions maintains Q34: What is the E value for the Q35: The SHE is assigned a voltage of Q36: What is the E0 value for the Q37: Single use, non-rechargeable batteries are referred to Q38: What is the E0 value for the Q40: The rusting witnessed in the sheet metal![]()
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents