In the natural radioactive decay chain 238 U undergoes a series of radioactive decays, finally producing the stable nuclide 206 Pb: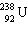
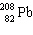 + A
+ A 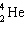 + B
+ B 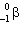 What are the values of A and B in this decay chain?
What are the values of A and B in this decay chain?
A) A = 10, B = 16
B) A = 8, B = 6
C) A = 6, B = 8
D) A = 16, B = 10
E) none of these
Correct Answer:
Verified
Q24: The nuclide 226 Ra undergoes a series
Q25: Provided in the table below are two
Q26: Provided in the table below are two
Q27: A sample that contains 5.7×10 - 9
Q28: Barium - 141 is a beta emitter.
Q30: Bismuth - 210, 210Bi, is an alpha
Q31: Consider comparing the two Phosphorous radionuclides given
Q32: When Plutonium - 239 changes to Uranium
Q33: A sample of a radioactive isotope decays
Q34: The nuclide that undergoes ![]()
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents