The graph here represents a p-V diagram where the x axis is the volume in units of 10-3 m3 and the y axis is the pressure in units of 105 N/m2. The work done by the ideal gas as it follows the path sequence (p1,V1) to (p2,V2) to (p3,V3) to (p1,V1) along the lines indicated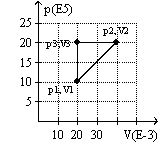
A) is 0 J.
B) is 10 kJ.
C) is -10 kJ.
D) cannot be determined from the given information.
Correct Answer:
Verified
Q11: A heat engine produces a power output
Q12: The efficiency of a heat engine
Q13: One-quarter of the energy a heat engine
Q14: The graph here represents a p-V diagram
Q15: The graph here represents a p-V diagram
Q17: The graph here represents a p-V diagram
Q18: Given a cyclic process on a p-V
Q19: In an adiabatic expansion of an ideal
Q20: In an adiabatic expansion of an ideal
Q21: During an isothermal process involving an ideal
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents