How many S 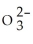 ions are contained in 99.6 mg of Na2SO3? The molar mass of Na2SO3 is 126.05 g mol-1.
ions are contained in 99.6 mg of Na2SO3? The molar mass of Na2SO3 is 126.05 g mol-1.
A) 1.52 × 1027 S 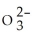 ions
ions
B) 4.76 × 1020 S 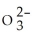 ions
ions
C) 2.10 × 1021 S  ions
ions
D) 1.05 × 1021 S 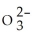 ions
ions
E) 9.52 × 1020 S 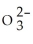 ions
ions
Correct Answer:
Verified
Q27: Calculate the mass percent composition of lithium
Q44: Give the mass percent of carbon in
Q82: How many molecules are in 114.86 g
Q86: How many molecules are in 1.500 g
Q94: How many molecules are in 0.1339 kg
Q101: Determine the molecular formula for a compound
Q102: Formic acid, HCOOH, is a major component
Q103: Formaldehyde, CH2O, is the simplest aldehyde and
Q112: How many atoms of carbon are contained
Q118: Hematite is the mineral form of iron(III)
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents