Choose the best Lewis structure for SF4.
A) 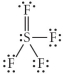
B) 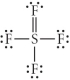
C) 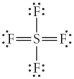
D) 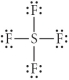
E) 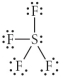
Correct Answer:
Verified
Q5: Choose the bond below that is LEAST
Q6: Identify the compound with the smallest dipole
Q7: Give the number of valence electrons for
Q8: Choose the best Lewis structure for CH2Cl2.
A)
Q9: Identify the compound with the highest percent
Q11: Identify the compound with the largest dipole
Q23: Identify the shortest bond.
A) single covalent bond
B)
Q26: Identify the number of bonding pairs and
Q33: Give the number of valence electrons for
Q39: Using periodic trends,place the following bonds in
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents