Draw the Lewis structure for NO2⁻ including any valid resonance structures.Which of the following statements is TRUE?
A) The nitrite ion contains one N-O single bond and one N  O double bond.
O double bond.
B) The nitrite ion contains two N-O bonds that are equivalent to 1 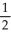 bonds.
bonds.
C) The nitrite ion contains two N  O double bonds.
O double bonds.
D) The nitrite ion contains two N-O single bonds.
E) None of the above are true.
Correct Answer:
Verified
Q19: Choose the best Lewis structure for OCl2.
A)
Q20: Choose the bond below that is MOST
Q21: Choose the best Lewis structure for PO43⁻.
A)
Q25: Choose the best Lewis structure for SeO42⁻.
A)
Q26: Which of the following resonance structures for
Q27: Draw the Lewis structure for CO32- including
Q29: Give the number of valence electrons for
Q35: Identify the strongest bond.
A) single covalent bond
B)
Q37: Draw the best Lewis structure for CH3-1.What
Q47: Using Lewis structures and formal charge,which of
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents