In this electrochemical cell,the reduction half reaction is 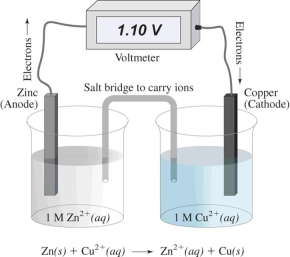
A) Cu2+(aq) + 2 e¯ → Cu(s)
B) Zn(s) → Zn2+(aq) + 2 e¯
C) Zn(s) → Cu(s)
D) Cu2+(aq) → Zn2+(aq)
Correct Answer:
Verified
Q6: During the chemical reaction in an electrochemical
Q10: In general,a modern hybrid vehicle is less
Q14: Whenever a substance is oxidized,
A)it is called
Q15: Which is not a necessary consideration for
Q16: In a voltaic cell,
A)oxidation takes place at
Q17: The current through a wire is most
Q20: The opposite of a galvanic cell is
A)a
Q21: Which is not a current or planned
Q22: The Prius,a hybrid car produced by Toyota,uses
Q23: Sunlight (solar radiation) may be turned directly
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents