How does the reaction of hydrogen and oxygen to produce energy in a fuel cell differ from their interaction during the direct combustion of hydrogen and oxygen? 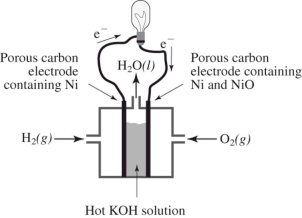
I.The direct combustion of hydrogen and oxygen produces several different products,whereas the fuel cell produces only water.
II.Much less heat energy is produced in a fuel cell than via direct combustion of hydrogen and oxygen.
III.In the fuel cell,there is an oxidation-reduction reaction between hydrogen and oxygen.In the direct combustion of hydrogen and oxygen,there is no such reaction.
IV.It is much easier to control the hydrogen and oxygen during direct combustion than during their reaction in a fuel cell.
A) I and II only
B) I,II and III only
C) I,II,III and IV
D) I and III only
Correct Answer:
Verified
Q7: Which of the following statements is not
Q25: Semiconductors are more effective than metals as
Q34: How does using hydrogen as the fuel
Q35: Which has not been suggested as a
Q36: A fuel cell does not "run down"
Q38: At present,it will be difficult and perhaps
Q40: As fuel cells become more widely accepted
Q42: Batteries must be used in addition to
Q43: Among the alternative sources for generating electricity
Q44: Which is not a valid argument supporting
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents