A NiCd battery uses nickel and cadmium to produce a potential difference. Using these equations, answer the following questions.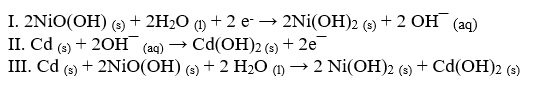
-Which equation best represents the reduction half-reaction?
A) I
B) II
C) III
D) I and II
Correct Answer:
Verified
Q9: Why doesn't water naturally become hydrogen and
Q33: Which argument(s) for the increasing use of
Q35: Which increases the efficiency of a photovoltaic
Q51: A NiCd battery uses nickel and cadmium
Q53: In the lead-acid (storage)battery, Q56: A NiCd battery uses nickel and cadmium Q57: Which is not a valid argument supporting Q58: In this photovoltaic (solar)cell, Q59: In a solar cell,placing semiconductors of the Q60: In recently designed fuel cells the corrosive![]()
![]()
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents