The reaction for the combustion of heptane is 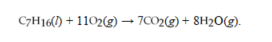 . How many liters of CO2 at STP are produced from the complete combustion of 2.00 moles of heptane?
. How many liters of CO2 at STP are produced from the complete combustion of 2.00 moles of heptane?
A) 44.8 L
B) 22.4 L
C) 157 L
D) 246 L
E) 314 L
Correct Answer:
Verified
Q26: According to the IUPAC convention,alkyl group names
Q38: What is the IUPAC name for this
Q39: What is the IUPAC name of the
Q40: According to the IUPAC convention for chemical
Q41: Which of the following compounds could have
Q44: A hydrocarbon with a double bond is
Q44: What is the IUPAC name of this
Q45: The complete combustion of 1 mole of
Q46: An alkyne is an organic compound that
Q52: What is the name for a three-carbon
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents