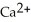 (aq)+ 2Na (s)→ Ca (s)+
(aq)+ 2Na (s)→ Ca (s)+ 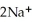 (aq)is a spontaneous reaction.
(aq)is a spontaneous reaction.
Activity Series = 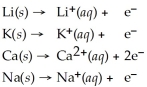
Correct Answer:
Verified
Q25: The driving force that causes electrons to
Q26: Gold metal is found at the bottom
Q27: Alkaline batteries are used in automobiles.
Q28: The cathode is the electrode at which
Q29: Most acids dissolve metals by the oxidation
Q31: A galvanic cell is a spontaneous electrochemical
Q32: Given that the Activity Series shown below
Q33: A spontaneous redox reaction can be used
Q34: An electrochemical cell is based on the
Q35: Electrolysis is used to recover many metals
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents