How would you expect the molecule 1,10-phenanthroline (shown below) to function as a ligand? 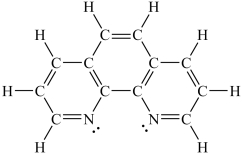
A) It would be expected to be a monodentate ligand.
B) It would be expected to be a bidentate ligand.
C) It would be expected to be a tridentate ligand.
D) It would be expected to be a tetradentate ligand.
E) It would not be expected to function as a ligand.
Correct Answer:
Verified
Q1: The electron configuration of a Ti atom
Q2: The electron configuration of an Fe2+ ion
Q5: The total number of electrons in the
Q8: The total number of electrons in the
Q13: The total number of electrons in the
Q23: Which response gives the correct coordination number
Q28: In the complex ion [Co(en)2Br2]+, the oxidation
Q29: Give the coordination number (C.N.) and oxidation
Q36: The correct formula for the dichlorobis(ethylenediamine)chromium(III) ion
Q48: Which of these complex ions would absorb
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents