A voltaic cell is made by placing an iron electrode in a compartment in which the Fe2+ concentration is 2.0 × 10-5 M and by placing a Pt electrode in the other compartment,in which the H+ concentration is 3.4 M and 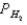 = 1.00 atm.The Fe2+/Fe half-cell reduction potential is -0.41 V,and the H+/H2 half-cell reduction potential is 0.00 V.What is the value of E° for this cell,and which electrode is the anode?
= 1.00 atm.The Fe2+/Fe half-cell reduction potential is -0.41 V,and the H+/H2 half-cell reduction potential is 0.00 V.What is the value of E° for this cell,and which electrode is the anode?
A) 0.41 V,Fe
B) 0.90 V,Pt
C) -0.41 V,Pt
D) -0.41 V,Fe
E) 0.41 V,Pt
Correct Answer:
Verified
Q68: Calculate the solubility product of silver iodide
Q69: Given: Q70: Which of the following statements is true Q71: What is the equilibrium constant (K)at 25°C Q72: For a certain reaction,ΔHº = -75.4kJ and Q74: A voltaic cell is made by placing Q75: Which of the following statements is true Q76: For a reaction in a voltaic cell,both Q77: If E°cell for a certain reaction is Q78: If the value of E°cell is 2.10![]()
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents