A voltaic cell is made by placing an iron electrode in a compartment in which the Fe2+ concentration is 2.0 × 10-5 M and by placing a Pt electrode in the other compartment,in which the H+ concentration is 3.4 M and 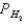 = 1.00 atm.The Fe2+/Fe half-cell reduction potential is -0.41 V,and the H+/H2 half-cell reduction potential is 0.00 V.What is E for the cell at equilibrium?
= 1.00 atm.The Fe2+/Fe half-cell reduction potential is -0.41 V,and the H+/H2 half-cell reduction potential is 0.00 V.What is E for the cell at equilibrium?
A) -0.41 V
B) 0.41 V
C) 0.69 V
D) 0.00 V
E) 0.28 V
Correct Answer:
Verified
Q95: What is the value of the reaction
Q96: The following cell is initially at standard-state
Q97: Which of the following statements is true
Q98: For the cell reaction
2MnO4- (aq)+ 5SO2(g)+ 2H2O(l)
Q99: What is the reduction potential for the
Q101: When Au is obtained by electrolysis from
Q102: Copper is electroplated from CuSO4 solution.A constant
Q103: In the electrolysis of an acid solution,oxygen
Q104: When an aqueous solution of AgNO3 is
Q105: How many faradays are involved in the
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents