For the reaction 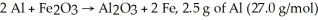
And 7.2 g of Fe2O3 (159.8 g/mol) produce how many g of Fe (55.9 g/mol) ?
A) 2.5 (55.9/27.0) g
B) 2.5 (55.9) (2) /(27.0) (2) g
C) 7.2 (55.9) (2) /159.8 g
D) 7.2 (55.9/159.8) g
E) 2.5 (55.9/159.8) g
Correct Answer:
Verified
Q13: What is the sum of the coefficients
Q14: The reactant that is in excess determines
Q15: The numbers in front of formulas in
Q16: Stoichiometric factor relates the amounts,in moles,of any
Q17: Which metal will produce the most hydrogen
Q19: When the equation K2S2O3 + I2 →
Q20: What is the sum of the coefficients
Q21: What volume of 2.0 M HCl,in mL,is
Q22: Potassium superoxide (KO2)can simulate a plant-type action
Q23: If 5.97 mL of a solution of
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents