Gases emitted during volcanic activity often contain high concentrations of hydrogen sulfide and sulfur dioxide.These gases may react to produce deposits of sulfur according to the equation: 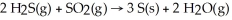
For the complete reaction of 6.41 mol of hydrogen sulfide:
A) 308 g of sulfur is formed
B) 410 g of sulfur dioxide is consumed
C) 231 g of water vapor is produced
D) 320 g of total products result
E) 628 g of total reactants are consumed
Correct Answer:
Verified
Q33: 24.0 g of ethane (C2H6)are burned to
Q34: Iron metal reacts with chlorine gas as
Q35: What mass of trisodium phosphate is required
Q36: For the reaction symbolized as HCl(aq)+ NaOH(aq)→
Q37: How many mL of 0.024 M solution
Q39: What is the molarity of 10.9 g
Q40: What volume of concentrated acetic acid
Q41: Consider the gaseous reaction:
N2H4(g)+ 3 O2(g)→ 2
Q42: How many grams of CrSO4 will be
Q43: The Haber Process for the production of
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents