The following nuclear reaction is known to occur:
2 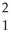 H →
H → 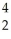 He
He
This reaction is used in a fusion bomb or H bomb.How many joules of energy are produced by this reaction if 33.0 g of 2H are used? You are given the following masses: electron 0.00055 amu;proton 1.00782 amu;neutron 1.00867 amu;2H atom 2.010410 amu; 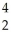
He atom 4.00260 amu.
A) 3.82 × 10-12 J
B) 2.30 × 1012 J
C) 3.77 × 1013 J
D) 7.60 × 1013 J
E) 1.34 J
Correct Answer:
Verified
Q23: The half-life of 238U,which decomposes to 206Pb,is
Q24: Choose the possible products for the nuclear
Q25: Most periodic tables indicate the elements for
Q26: What is the decay constant for 131I
Q27: There have been two ways of making
Q29: Choose the INCORRECT statement.
A)Bi-209 has the highest
Q30: Transuranium elements can be produced by bombarding
Q31: The charcoal from ashes found in a
Q32: Which of the following types of nuclear
Q33: The observation that a 30 g sample
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents