Ibuprofen is used as an analgesic for the relief of pain, and also to help reduce fever.What is the hybridization state of carbon indicated by the arrow in the structure of ibuprofen shown below? 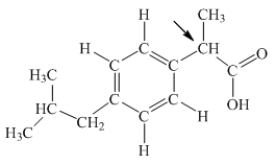
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
Correct Answer:
Verified
Q65: According to Valence Bond Theory, which orbital
Q66: What is the hybridization of the As
Q68: Indicate the type of hybrid orbitals used
Q72: The hybridization of the central nitrogen atom
Q72: N,N-diethyl-m-tolumide (DEET)is the active ingredient in many
Q74: N,N-diethyl-m-tolumide (DEET)is the active ingredient in many
Q79: According to Valence Bond Theory which orbital
Q80: Ibuprofen is used as an analgesic for
Q82: Consider the species Cl2+, Cl2, and Cl2-.Which
Q84: A sp2 hybridized terminal oxygen atom
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents