The equilibrium between carbon dioxide gas and carbonic acid is very important in biology and environmental science. CO2 (aq) + H2O (l)
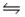
H2CO3 (aq)
Which one of the following is the correct equilibrium constant expression (Kc) for this reaction?
A) 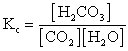
B) Kc = 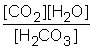
C) Kc = 
D) Kc = 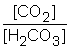
E) Kc = 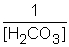
Correct Answer:
Verified
Q11: The brown gas NO2 and the colorless
Q12: Calculate Kp for the reaction 2NOCl(g)
Q13: Which is the correct equilibrium constant expression
Q14: When the following reaction is at equilibrium,
Q15: On analysis, an equilibrium mixture for the
Q17: When the following reaction is at equilibrium,
Q18: Given the following information: 2A(g)+ B(g)
Q19: Carbon tetrachloride reacts at high temperatures with
Q20: The reaction A(g)+ 2B(g) Q21: If one starts with pure NO2(g)at a![]()
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents