Consider the reaction N2(g)+ 3H2(g) 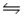
2NH3(g).If nitrogen is added to the system at equilibrium, what will happen to the ammonia concentration?
Correct Answer:
Verified
Q66: Consider the reaction, N2(g)+ 3H2(g) 
Q67: Consider the reaction N2(g)+ 3H2(g) 
Q68: Given the following data for the reaction:
Q69: Consider the reaction N2(g)+ 3H2(g) 
Q70: Consider the reaction N2(g)+ 3H2(g) 
Q72: Consider the reaction, N2(g)+ 3H2(g) 
Q73: A solution was prepared such that the
Q74: Given the following data for the reaction:
Q75: Consider the reaction N2(g)+ 3H2(g)
Q76: When the reaction 2H2S(g) ![]()
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents