Calcium carbonate decomposes at high temperatures to give calcium oxide and carbon dioxide as shown below.
CaCO3(s) 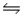
CaO(s)+ CO2(g)
The KP for this reaction is 1.16 at 800°C.A 5.00 L vessel containing 10.0 g of CaCO3(s)was evacuated to remove the air, sealed, and then heated to 800°C.Ignoring the volume occupied by the solid, what will be the overall mass percent of carbon in the solid once equilibrium is reached?
Correct Answer:
Verified
View Answer
Unlock this answer now
Get Access to more Verified Answers free of charge
Q83: What is the correct equilibrium constant expression
Q84: Consider the chemical reaction 2NH3(g) 
Q85: What conditions are used in the Haber
Q86: The Kc for the reaction CO2(g)+ H2(g)
Q87: Consider the following equilibrium,
4NH3(g)+ 3O2(g) 
Q89: Calcium carbonate decomposes at high temperatures to
Q90: Consider the following equilibrium,
4NH3(g)+ 3O2(g) 
Q91: Consider the following equilibrium,
4NH3(g)+ 3O2(g) 
Q92: The data below refer to the following
Q93: Consider the equilibrium C(s)+ H2O(g)
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents