Assuming a coordination complex is formed with Fe2+ and 1,10-phenanthroline (shown below) , which of the following statements is true? 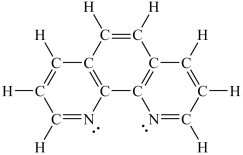
A) If two 1,10-phenanthroline molecules are coordinated to the iron ion, then the coordination number is 2.
B) If two 1,10-phenanthroline molecules are coordinated to the iron ion, then the coordination number is 6.
C) If three 1,10-phenanthroline molecules are coordinated to the iron ion, then the coordination number is 3.
D) If three 1,10-phenanthroline molecules are coordinated to the iron ion, then the coordination number is 6.
E) If four 1,10-phenanthroline molecules are coordinated to the iron ion, then the coordination number is 4.
Correct Answer:
Verified
Q2: The electron configuration of an Fe2+ ion
Q3: In Na3[Ni(SCN)5], how many 3d electrons does
Q5: The total number of electrons in the
Q12: In the complex ion [Fe(CN)6]4-, the oxidation
Q13: The total number of electrons in the
Q15: In K4[Fe(CN)6], how many 3d electrons does
Q16: The total number of electrons in the
Q19: Which of the following choices can function
Q20: The electron configuration of a Cr3+ ion
Q39: In the coordination compound [Cr(NH3)(en)2Cl]Br2, the coordination
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents