Ibuprofen is used as an analgesic for the relief of pain, and also to help reduce fever. 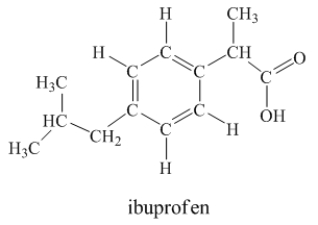 How many sigma bonds and pi bonds are contained in a ibuprofen molecule?
How many sigma bonds and pi bonds are contained in a ibuprofen molecule?
Correct Answer:
Verified
View Answer
Unlock this answer now
Get Access to more Verified Answers free of charge
Q67: Indicate the type of hybrid orbitals used
Q68: Indicate the type of hybrid orbitals used
Q78: The number of pi bonds in the
Q80: What is the hybridization of As in
Q83: In which of the following would the
Q84: In which of the following would the
Q96: In which of the following would the
Q106: Explain why CO2 is nonpolar, but OCS
Q112: Use VSEPR theory to predict the molecular
Q119: Using periodic trends, arrange the following molecules
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents