For the reaction SO2(g) + NO2(g) 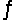 SO3(g) + NO(g) , the equilibrium constant is 18.0 at 1,200ºC. If 1.0 mole of SO2 and 2.0 moles of NO2 are placed in a 20.L container, what concentration of SO3 will be present at equilibrium?
SO3(g) + NO(g) , the equilibrium constant is 18.0 at 1,200ºC. If 1.0 mole of SO2 and 2.0 moles of NO2 are placed in a 20.L container, what concentration of SO3 will be present at equilibrium?
A) 0.48 mol/L
B) 0.11 mol/L
C) 0.95 mol/L
D) 2.22 mol/L
E) 18 mol/L
Correct Answer:
Verified
Q23: Consider the following equilibria: 2SO3(g) 
Q24: For the equilibrium reaction 2SO2(g)+ O2(g)
Q25: Hydrogen iodide decomposes according to the equation
Q26: At 400ºC, Kc = 64 for the
Q27: For the following reaction at equilibrium,
Q29: Consider the reaction N2(g)+ O2(g) 
Q30: For the following reaction at equilibrium
Q31: For the nitrogen fixation reaction 3H2(g)+ N2(g)
Q32: Consider the following reactions and their associated
Q33: For the reaction H2(g)+ I2(g) 
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents