Carbon-11 decays by positron emission: 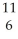 C →
C → 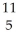 B +
B + 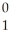 e The decay occurs with a release of 2.87 × 1011 J per mole of carbon-11. When 5.00 g of carbon-11 undergoes this radioactive decay, ________ g of mass is converted to energy.
e The decay occurs with a release of 2.87 × 1011 J per mole of carbon-11. When 5.00 g of carbon-11 undergoes this radioactive decay, ________ g of mass is converted to energy.
A) 1.45 × 10-3
B) 4.35 × 105
C) 1.45 × 10-6
D) 6.90 × 102
E) 1.59 × 10-2
Correct Answer:
Verified
Q126: Strontium-90 is a byproduct in nuclear reactors
Q127: 131I has a half-life of 8.04 days.
Q128: Potassium-40 decays to argon-40 with a half-life
Q129: A freshly prepared sample of curium-243 undergoes
Q130: The product of the nuclear reaction in
Q132: A rock contains 0.153 mg of lead-206
Q133: The decay of a radionuclide with a
Q134: Carbon-11 is used in medical imaging. The
Q135: If we start with 1.000 g of
Q136: The mass of a proton is 1.673
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents